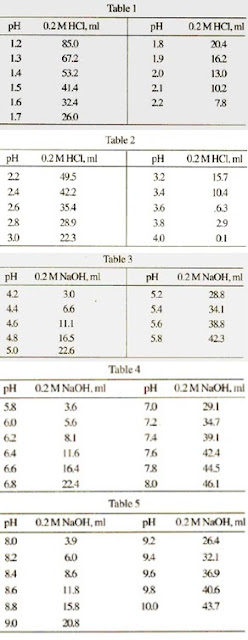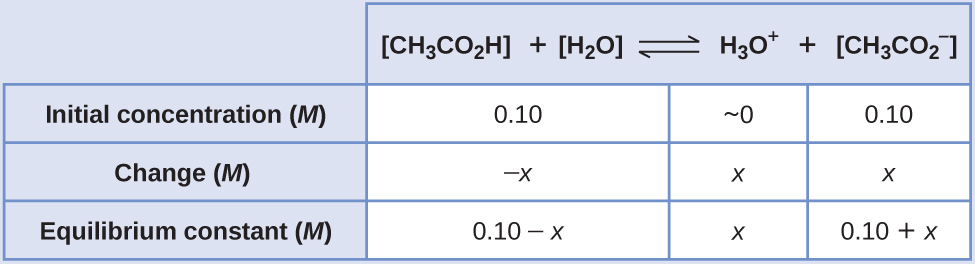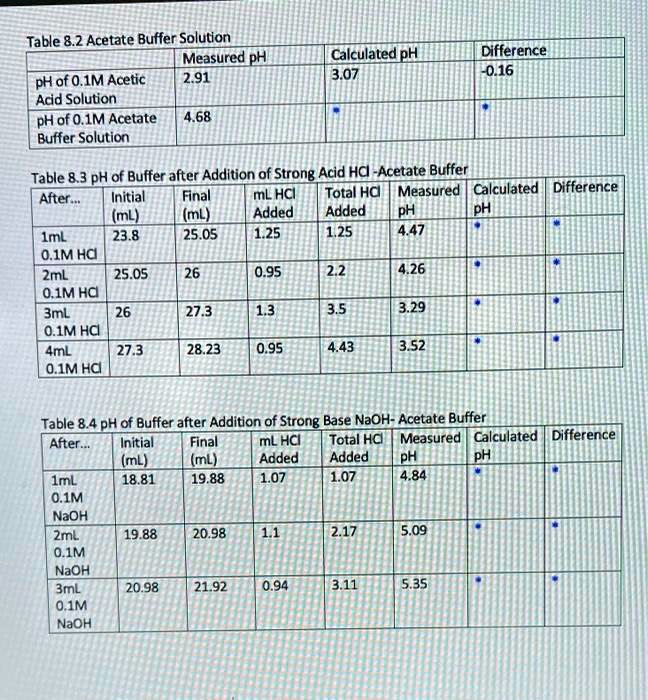
SOLVED: Table 8.2 Acetate Buffer Solytion Measured pH PH of 0.1M Acetic 2.91 Acid Solution pH of O.1M Acetate 4.68 Buffer Solution CalculatedpH 3.07 Difference 0.16 Table 8.3 pH of Buffer after

Breaking old habits: Moving away from commonly used buffers in pharmaceuticals - European Pharmaceutical Review
![PDF] LABORATORY TRIAL OF PROTEIN DETERMINATION IN URINE USING DIFFERENT PH VALUES OF ACETIC ACID AND ACETATE BUFFER METHOD | Semantic Scholar PDF] LABORATORY TRIAL OF PROTEIN DETERMINATION IN URINE USING DIFFERENT PH VALUES OF ACETIC ACID AND ACETATE BUFFER METHOD | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/4c8c15bf5aceadd13bacedde9640e7451f0516af/5-Table1-1.png)
PDF] LABORATORY TRIAL OF PROTEIN DETERMINATION IN URINE USING DIFFERENT PH VALUES OF ACETIC ACID AND ACETATE BUFFER METHOD | Semantic Scholar

Table II from Effect of Acetate and Carbonate Buffers on the Photolysis of Riboflavin in Aqueous Solution: A Kinetic Study | Semantic Scholar
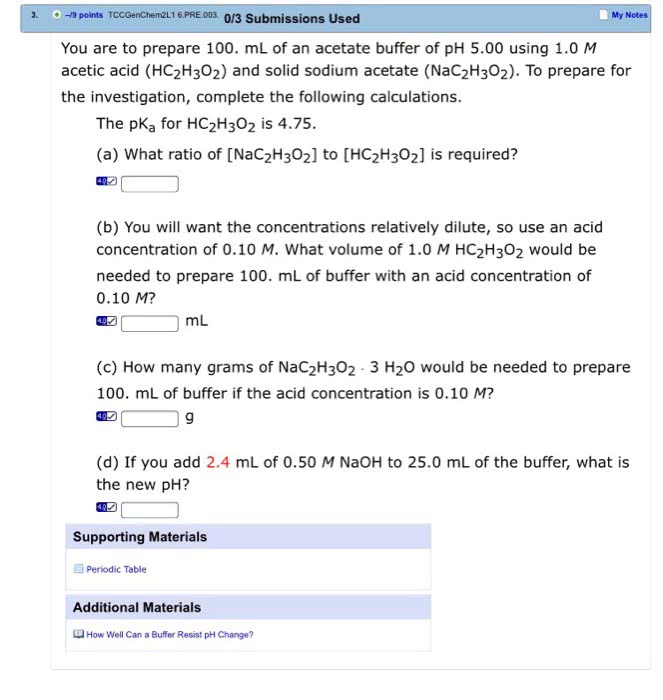
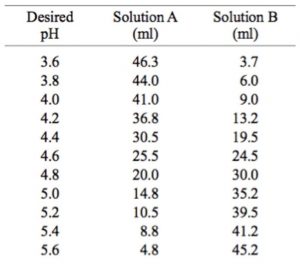
OneClass: You are to prepare 100. mL of an acetate buffer of pH 5.00 using 1.0 M acetic acid (HC_2H_3...

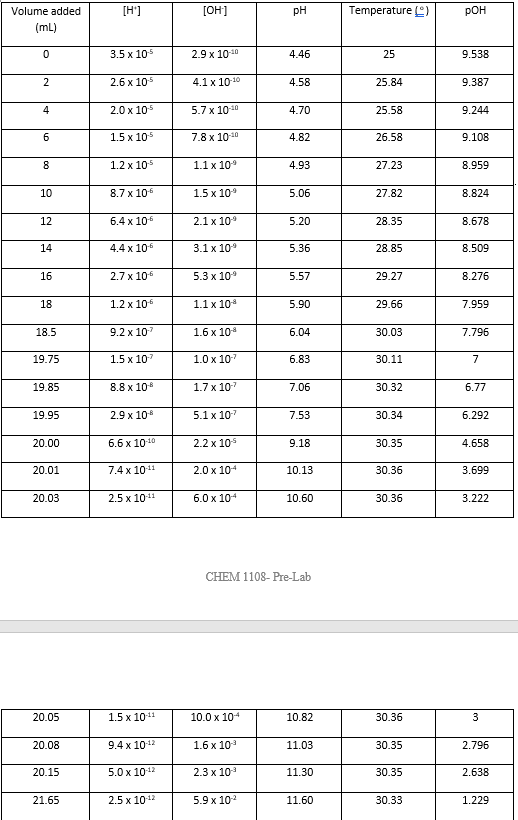
SOLVED: EXPERIMENT PREPARING A BUFFER Data Sheet Table 3: Sodium Acetate Data Sodium Acotate (g1 Molarity of Sodium Acetate (Step 4 Table 4: Buffer Solutions and pH Readings for Beakers A, B,€