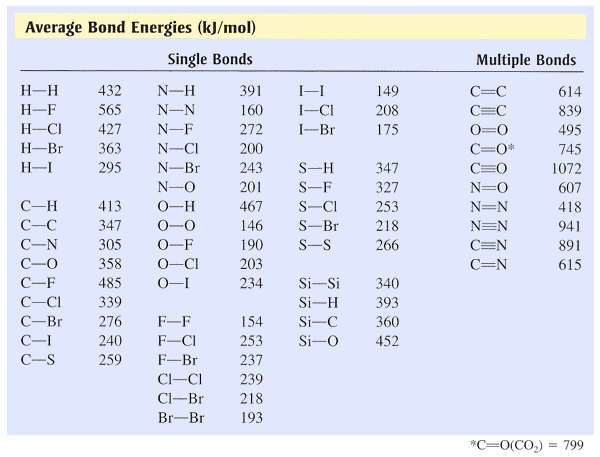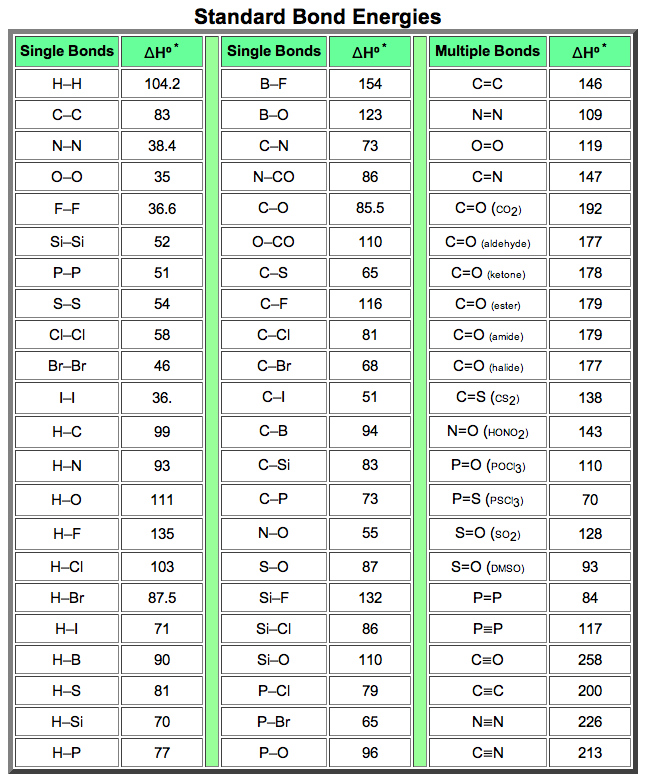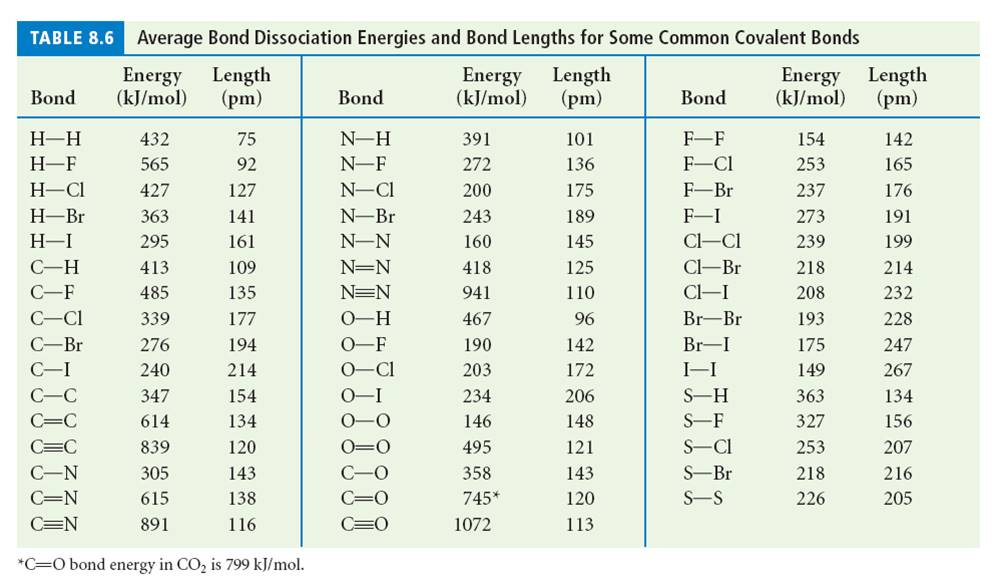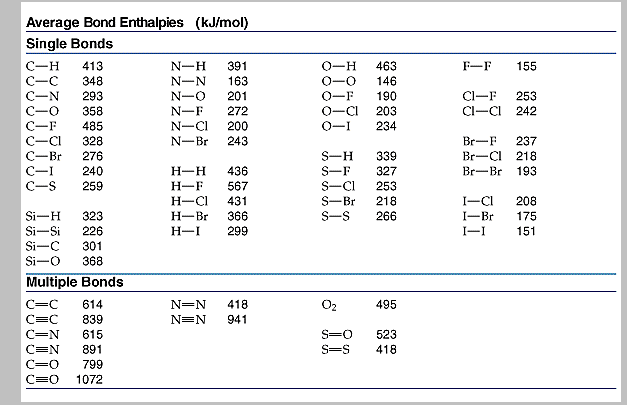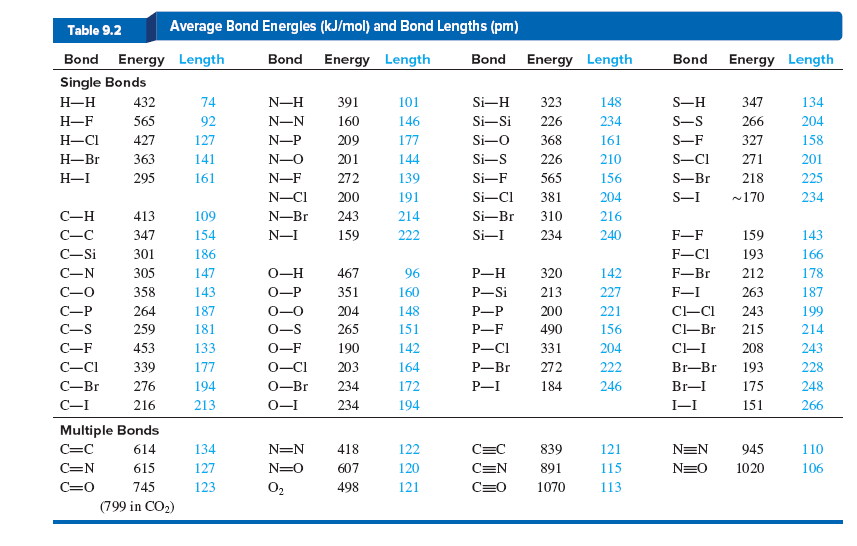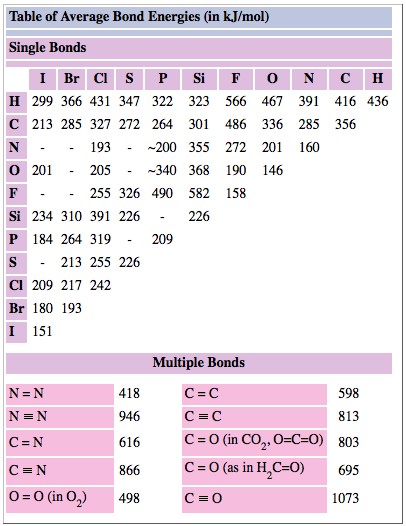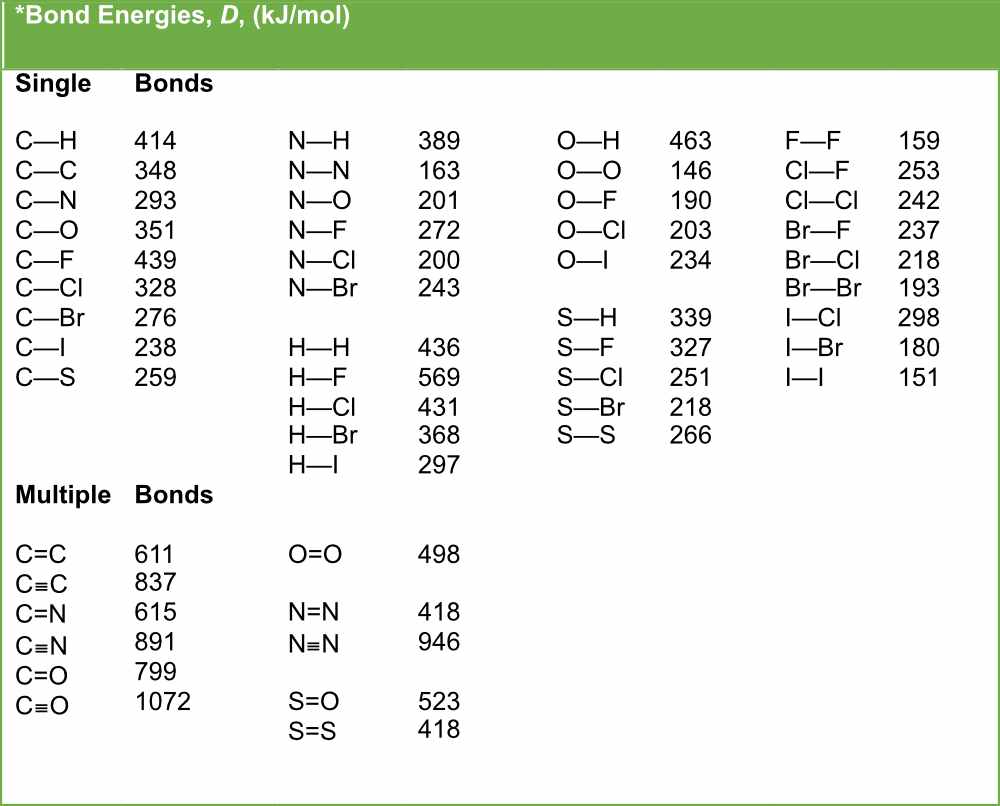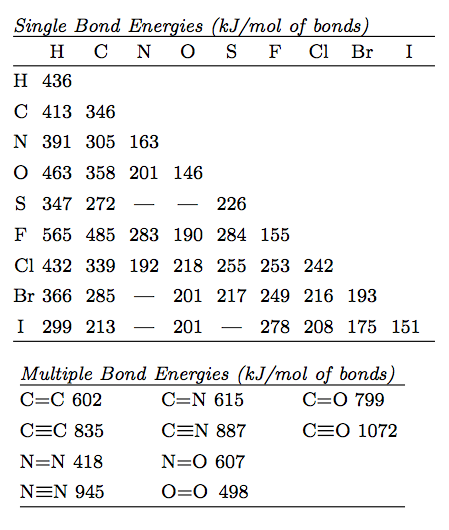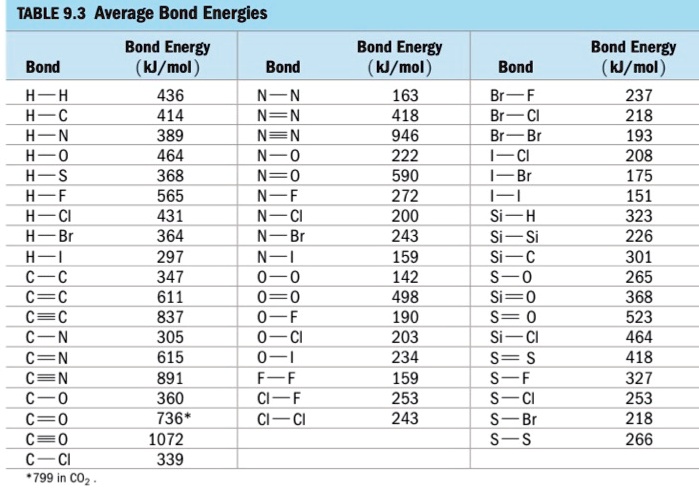
SOLVED: TABLE 9.3 Average Bond Energies Bond Energy Bond kJ/mol Bond 436 Bond Energy kJ/mol 163 418 946 222 590 272 200 243 159 142 498 190 203 234 159 253 243 Bond Energy kJ/ mol ) 237 218 193 208 175 151 323 226 301 265 368 523 464 418 327 253 218 ...

Bond Energy Table.pdf - Bond Energies Bond H–H H–C H–N H–O H–S H–F H–Cl H–Br H–I BE (kJ | Course Hero

Active Thermochemical Tables: Sequential Bond Dissociation Enthalpies of Methane, Ethane, and Methanol and the Related Thermochemistry | The Journal of Physical Chemistry A