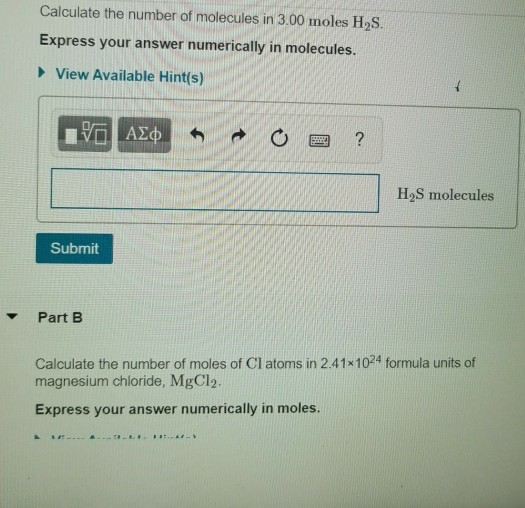
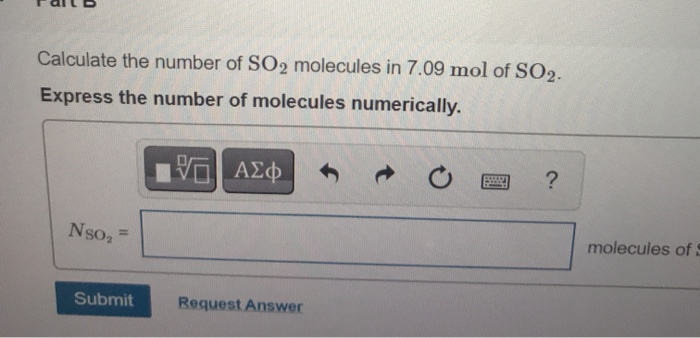
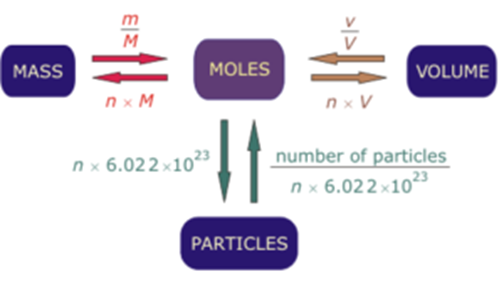
How to calculate the number of molecules of an element in a compound if I only know the total mass - Quora

Calculate the number of molecules in 36gm of H2o - Chemistry - Atoms and Molecules - 14431885 | Meritnation.com
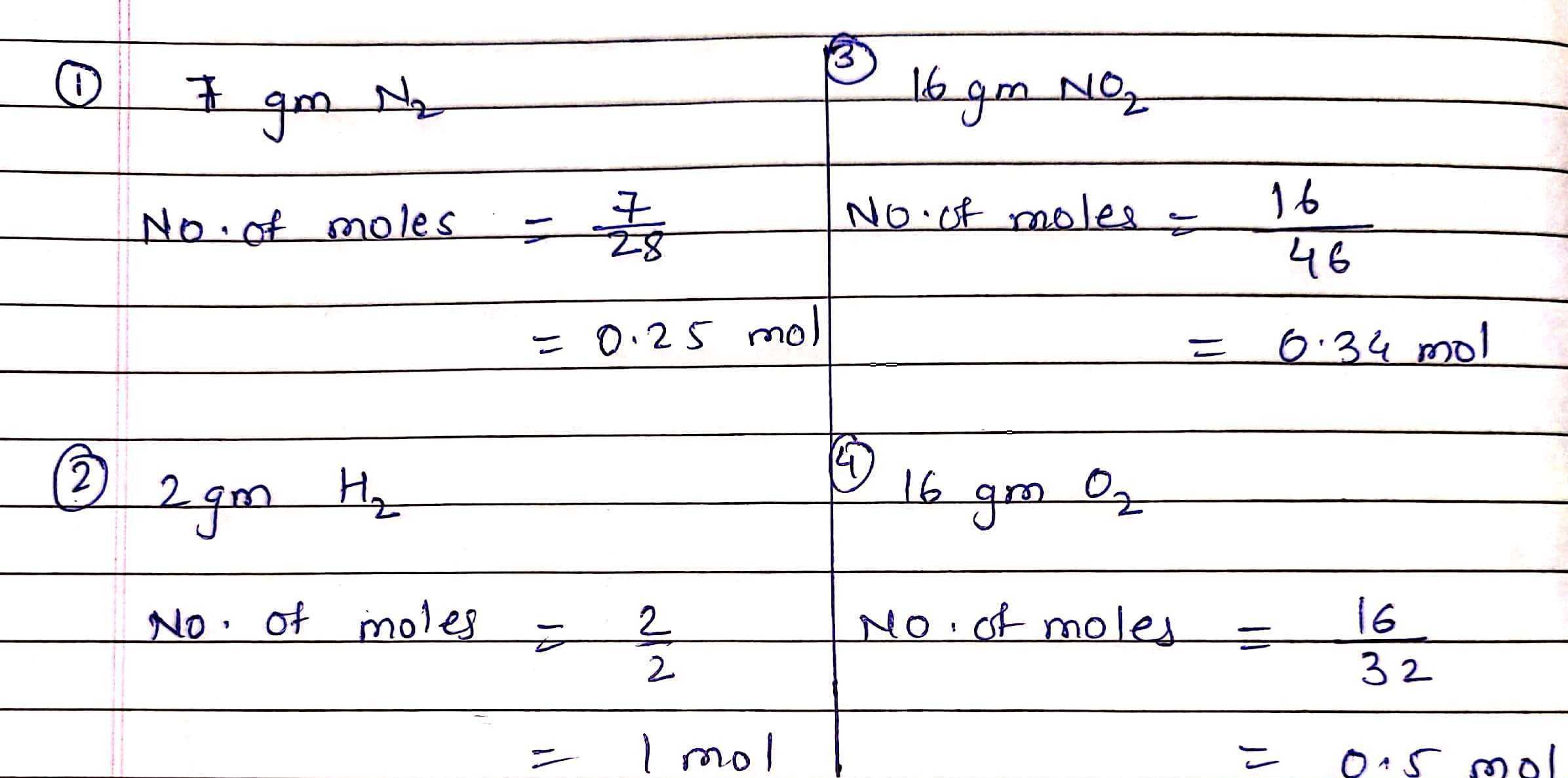
which has maximum number of molecules 1 7 gmn2 2 2 gmh2 3 16 gmno2 4 16 gmo2 8rxwz5pp -Chemistry - TopperLearning.com

SOLVED: Calculate the number of molecules present in each of the following samples. a. 6.37 mol of carbon monoxide b. 6.37 g of carbon monoxide c. 2.62 × 10^-6 of water d.

Calculate the number of molecules of sulphur (S8) present in 16 g of solid sulphur (Aton mass of S = 32 )

Calculate the number of molecules in 2 xx 10^(-6)m^(3) of a perfect gas at 27^(@) C and at a pressure of 0.01 mm of mercury. Mean KE of a molecules at 27^(@)

SOLVED:Calculate the number of molecules present in each of the following samples. a. 6.37 mol of carbon monoxide b. 6.37 g of carbon monoxide c. 2.62 ×10^-6 of water d. 2.62 ×10^-6