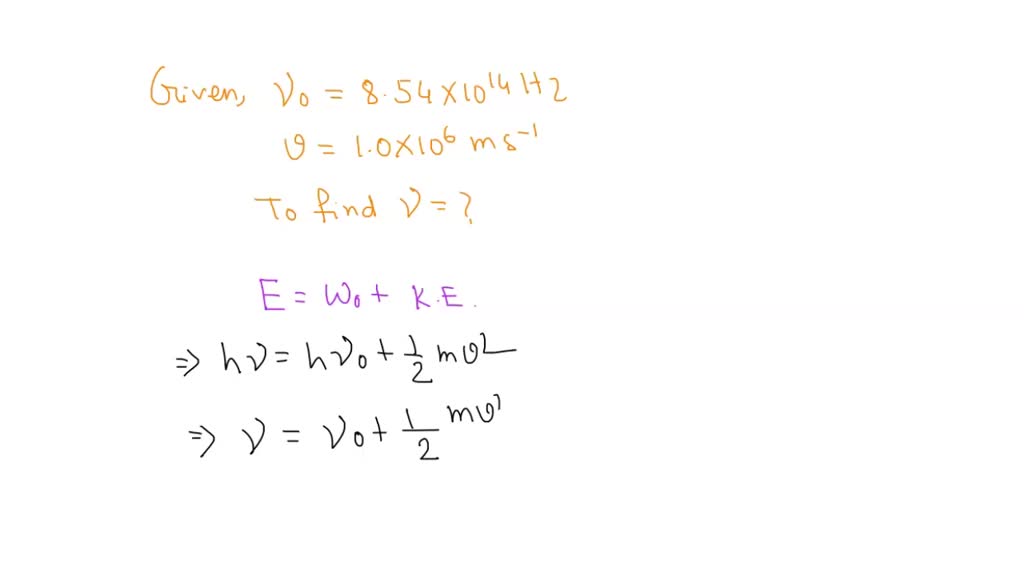Calculate the threshold frequency of photon for photoelectric emission from a metal of work - YouTube

Electrons are emitted with zero velocity from metal surface when it is exposed to radiation of wave¬length 6800 A. Calculate threshold frequency (v0) and work function (W0) of the metal - CBSE

Electrons are emitted with zero velocity from a metal surface when it is exposed to radiation of wavelength 6800 ∘A . Calculate threshold frequency v0 and work function w0 of the metal.

The work function of Cs is 2.14eV.Find (a) threshold frequency for Cs (b) Wavelength of incident - YouTube

SOLVED: The threshold frequency for zinc metal is 1.18 x 1ol5 s-1 . What is the energy in joules of a photon of this radiation? b) Calculate the kinetic energy of the

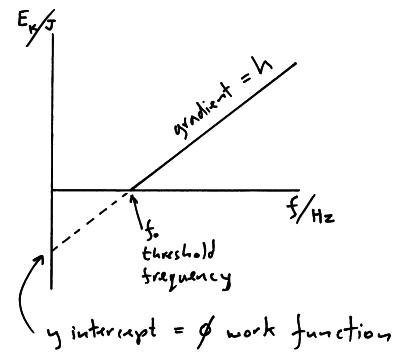
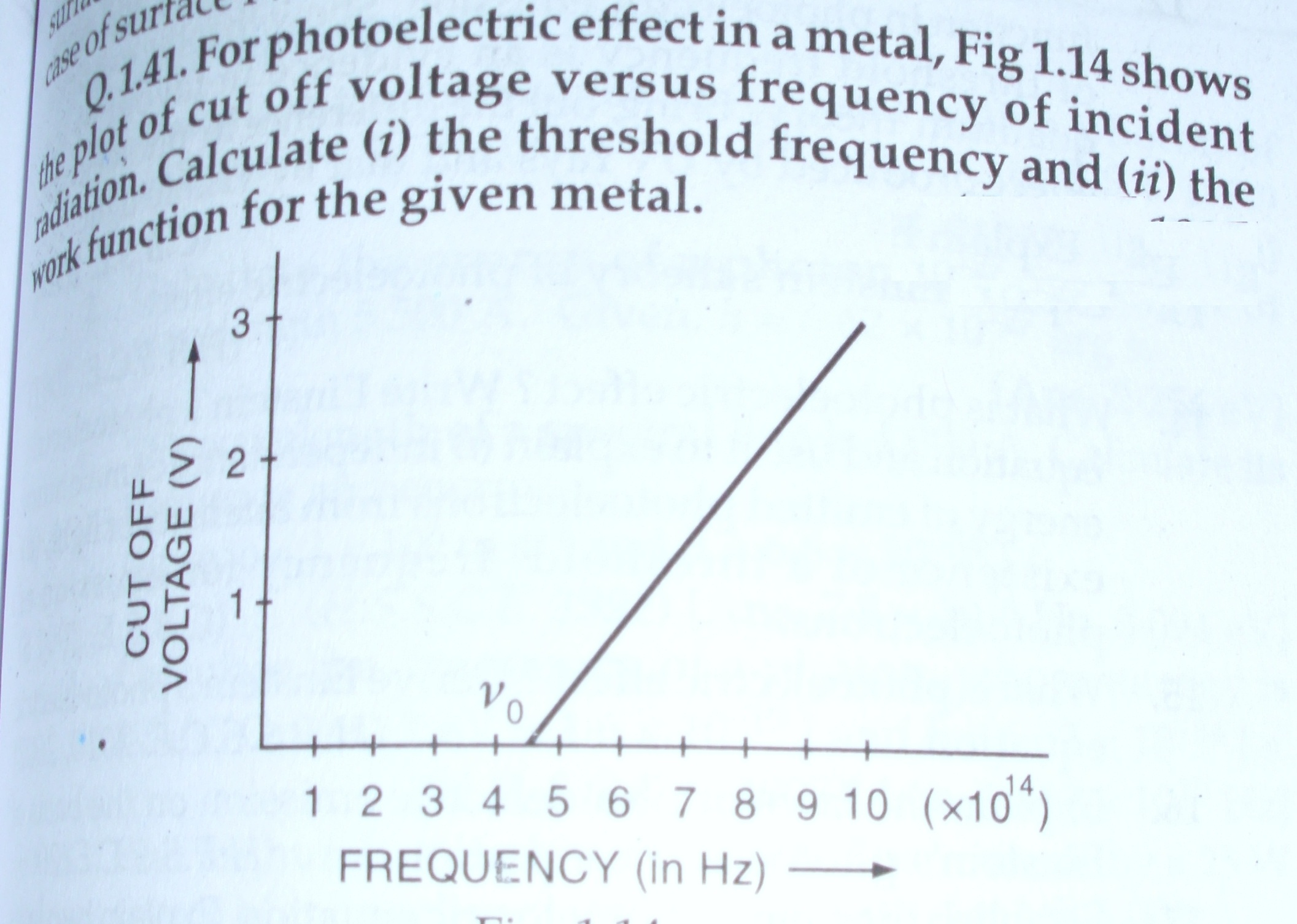
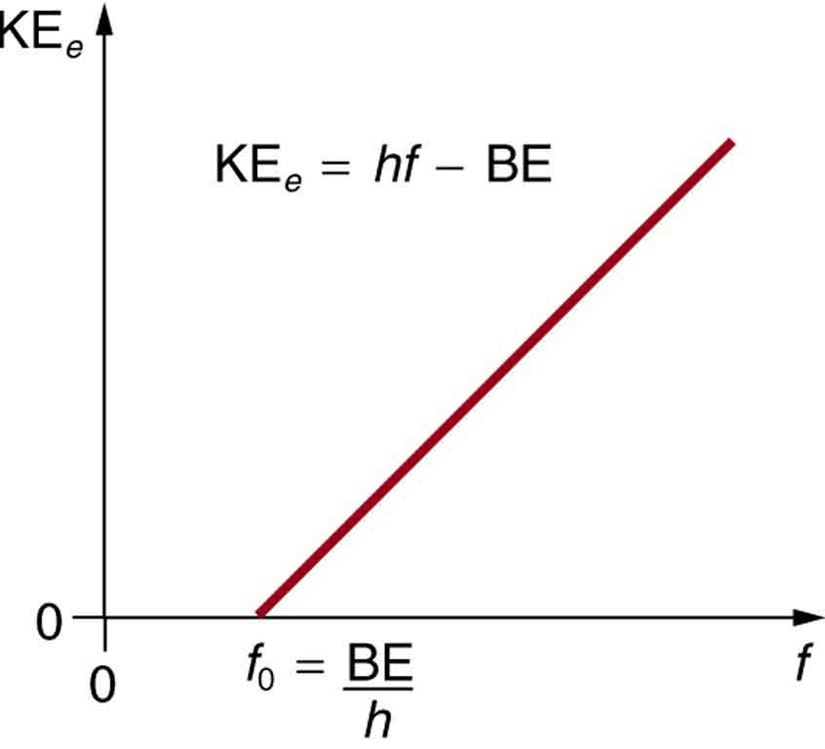
SOLVED: Calculate the threshold energy in kJ/mol of electrons in a metal, given that the lowest frequency photon for which the photoelectric effect is observed is 8.99 × 1014 Hz.. A. 394

The threshold frequency v0 for a metal is 7.0 × 10^14 s ^-1. Calculate the kinetic energy of an electron emitted when radiation of frequency v = 1.0 × 10^15 s^-1 hits the metal.

Calculate the threshold frequency of the metal from which the photoelectrons are emitted with zero velocity when exposed to radiation - Chemistry - - 14932835 | Meritnation.com

The threshold frequency v0 for a metal is 7.0 × 10^14 s ^-1. Calculate the kinetic energy of an electron emitted when radiation of frequency v = 1.0 × 10^15 s^-1 hits the metal.
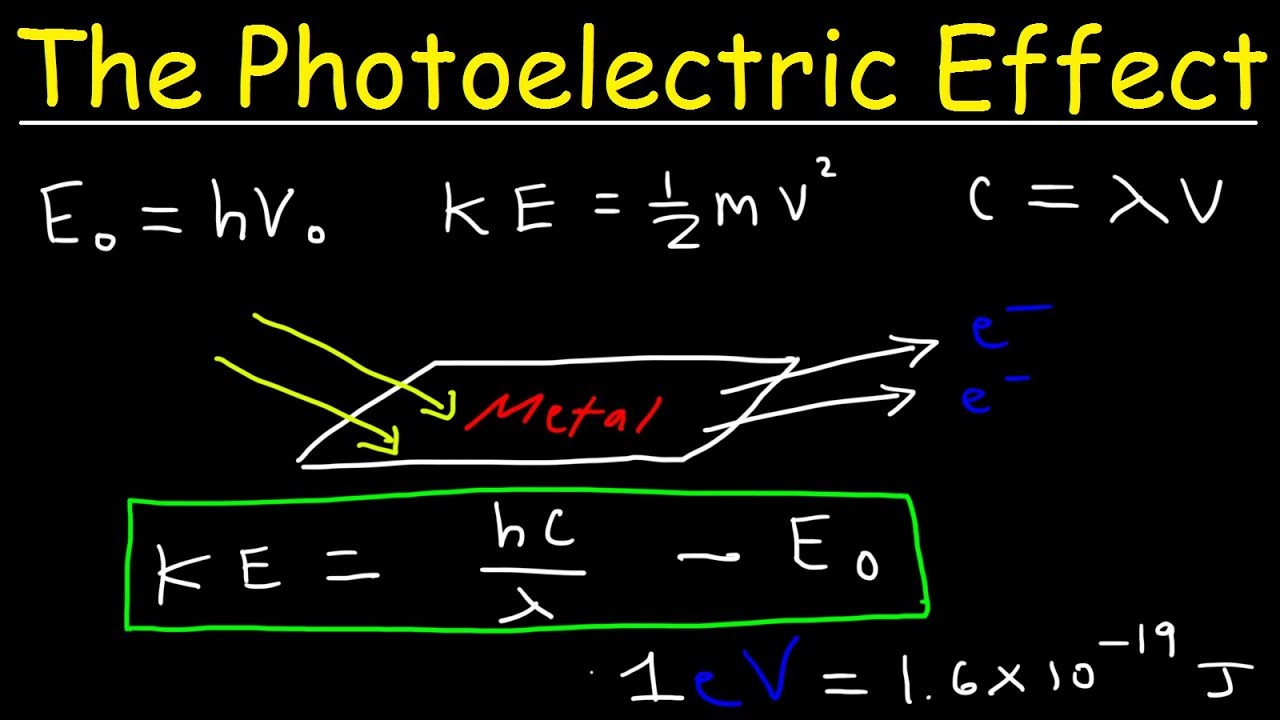
Photoelectric Effect, Work Function, Threshold Frequency, Wavelength, Speed & Kinetic Energy, Electr - YouTube
calculate the threshold frequency of the metal from which the photoelectrons are emitted with zero velocity when exposed to radiation of wavelength 6800 Armstrong