Open Letter to Gavi, vaccine manufacturers and donor governments ahead of the Global Vaccination Summit in Brussels | Médecins Sans Frontières Access Campaign

Preclinical in vitro and in vivo profile of a highly-attenuated, broadly efficacious pneumolysin genetic toxoid - ScienceDirect

Resurgence of pneumococcal meningitis in Europe and Northern America - Clinical Microbiology and Infection

Safety and Immunogenicity of a Candidate Bioconjugate Vaccine against Shigella flexneri 2a Administered to Healthy Adults: a Single-Blind, Randomized Phase I Study | Clinical and Vaccine Immunology

Polysaccharide conjugate vaccine: A kind of vaccine with great development potential - ScienceDirect
Improving vaccines against <italic toggle='yes'>Streptococcus pneumoniae</italic> using synthetic glycans
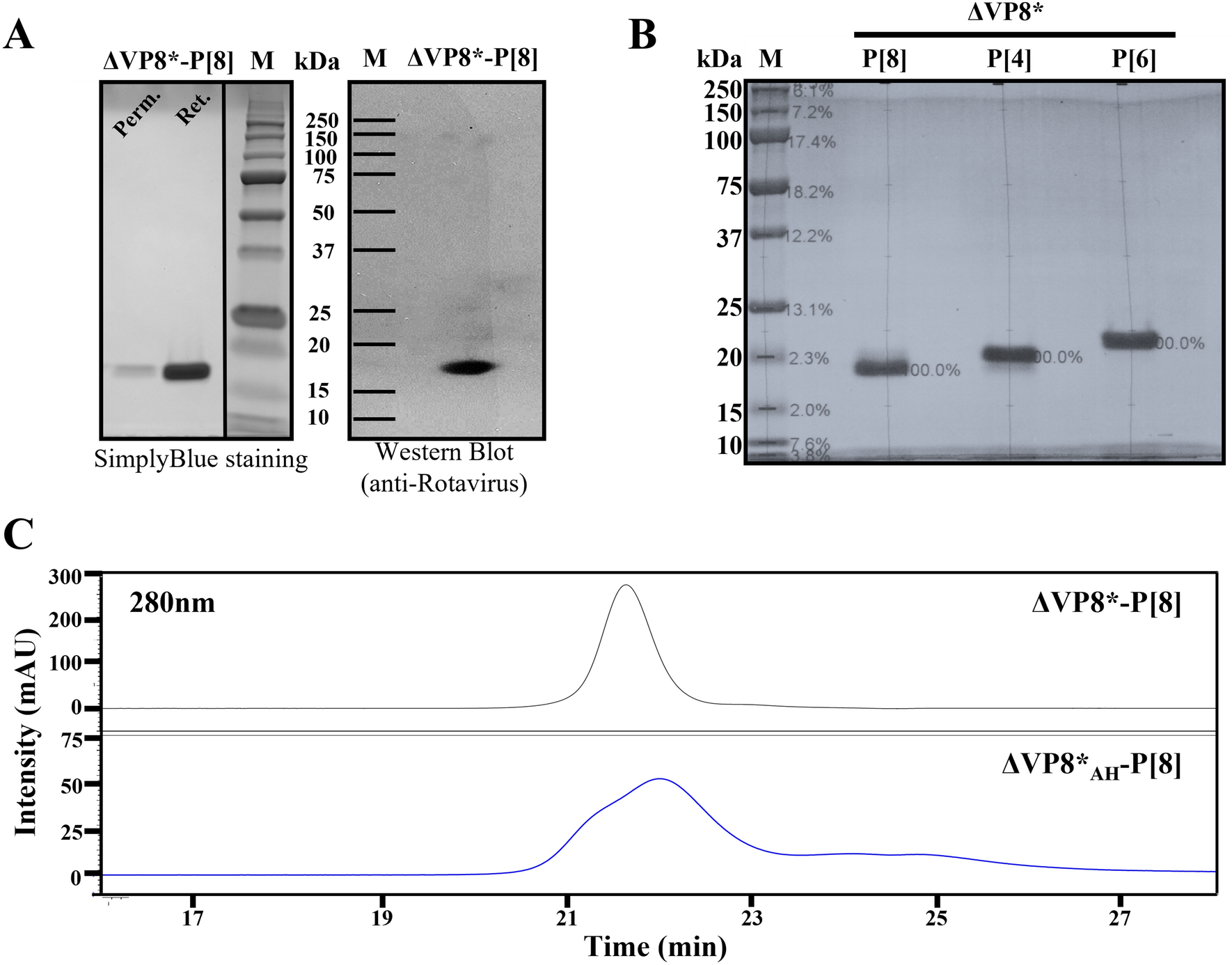
Rotavirus spike protein ΔVP8* as a novel carrier protein for conjugate vaccine platform with demonstrated antigenic potential for use as bivalent vaccine | Scientific Reports

Enabling Lyophilized Pneumococcal Conjugate Vaccines Through Formulation Design and Excipient Selection Suitable for A Multivalent Adjuvanted Vaccine - Journal of Pharmaceutical Sciences
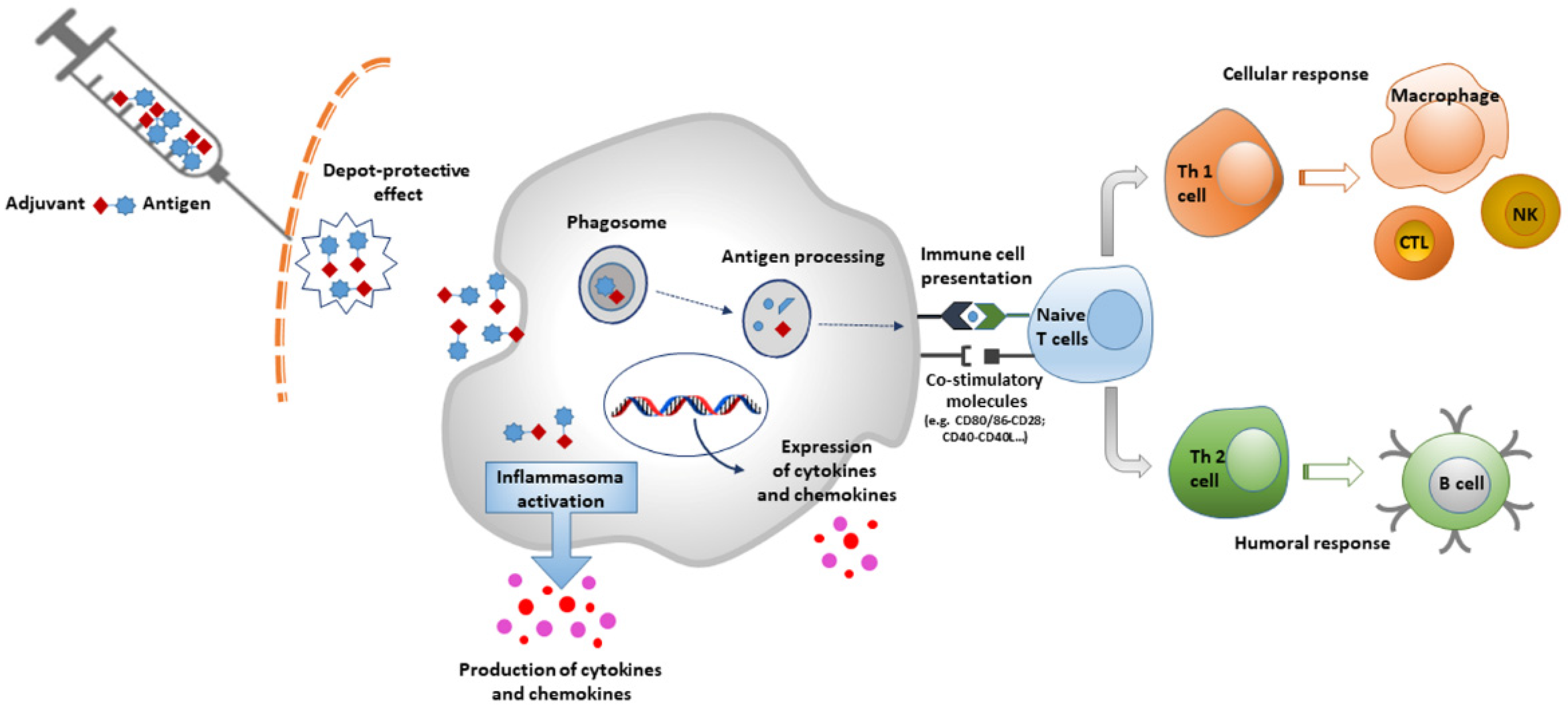
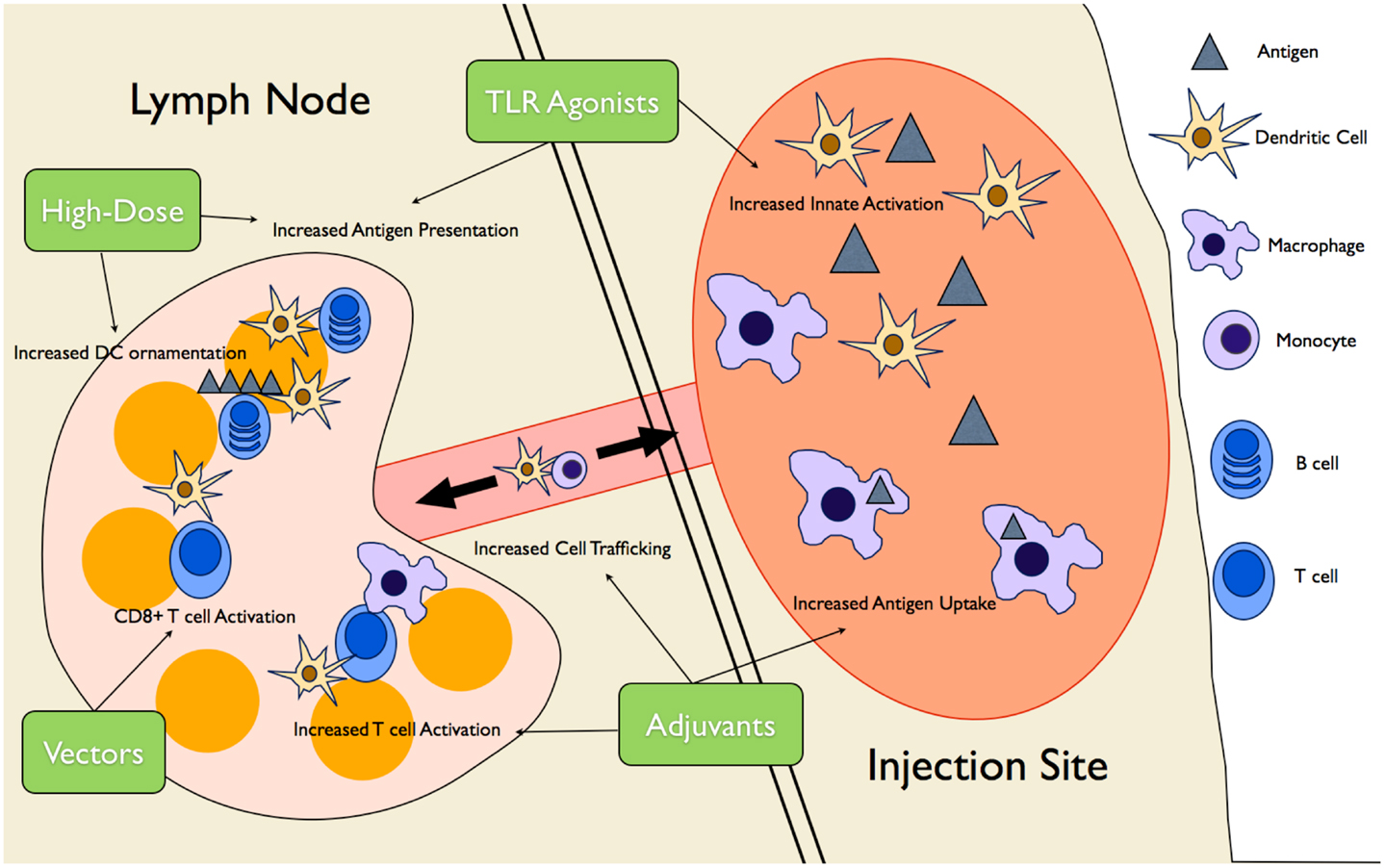
Vaccines | Free Full-Text | An Overview of Vaccine Adjuvants: Current Evidence and Future Perspectives | HTML
Certificat de înregistrare al medicamentului - nr. 21663 din 23.04.2015 Anexa 1 Prospect: Informaţii pentru utilizator Synflor

Developing vaccines in the era of genomics: a decade of reverse vaccinology - Clinical Microbiology and Infection