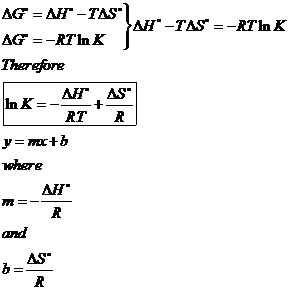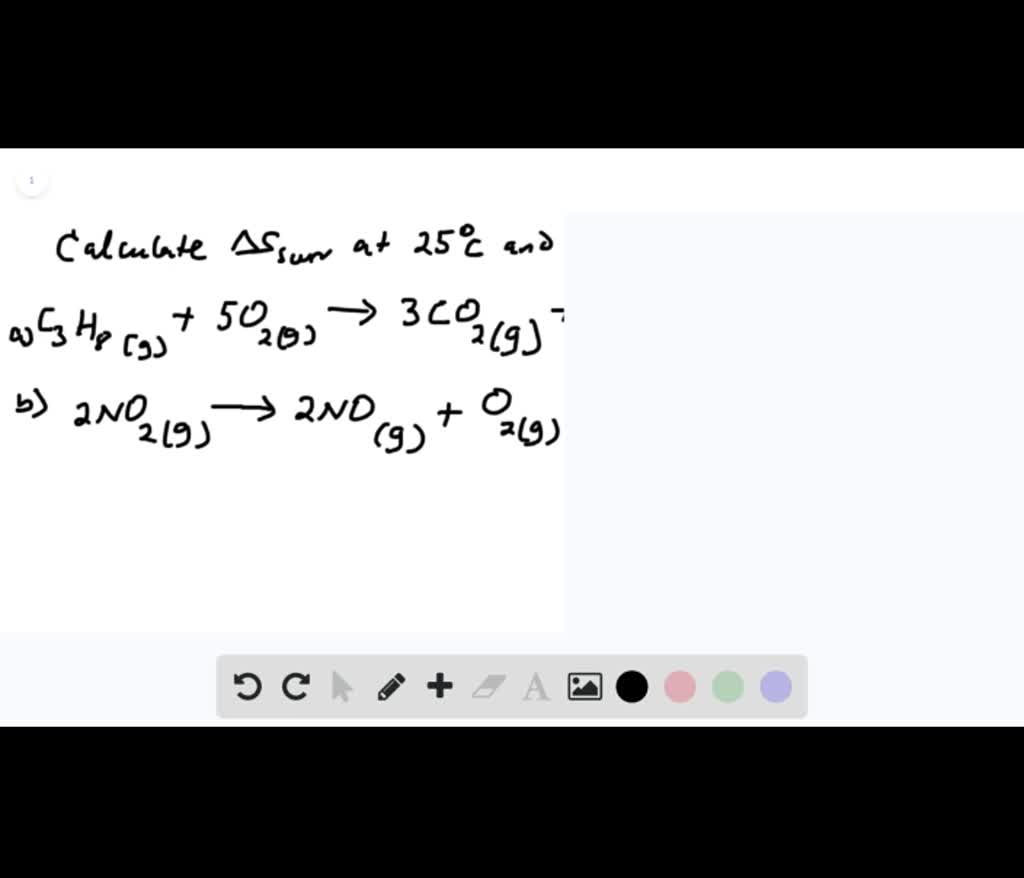
SOLVED:Calculate ΔS surr for the following reactions at 25^∘ C and 1 atm . a. C3 H8(g)+5 O2(g) ⟶3 CO2(g)+4 H2 O(l)ΔH^∘=-2221 kJ b. 2 NO2(g) ⟶2 NO(g)+O2(g) ΔH^ρ=112 kJ

Calculate ΔS univ (in J/K) for the chemical reaction: C(graphite) + 2H2(g)→CH4(g);ΔH300^o = - 75.0kJ . The standard entropies of C(graphite),H2(g),CH4(g) are 6.0,130.6 and 186.2J/K - mol , respectively.

physical chemistry - Why doesn't Delta S total = 0 for this reversible process? - Chemistry Stack Exchange

SOLVED:Predict the sign of ΔS^∘ and then calculate ΔS^∘ for each of the following reactions. a. H2(g)+(1)/(2) O2(g) ⟶H2 O(l) b. 2 CH3 OH(g)+3 O2(g) ⟶2 CO2(g)+4 H2 O(g) c. HCl(g) ⟶H^+(a q)+
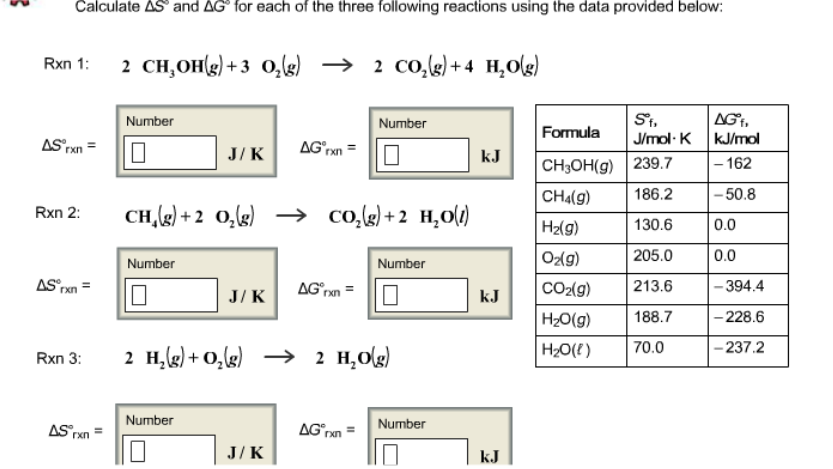
![Calculate deltaS and deltaG for each of t [{Image src='img236741055943655201772.jpg' alt='' caption=''}]he following 3 rxns using the data provided. Calculate delta S and delta G for each of the three | Homework.Study.com Calculate deltaS and deltaG for each of t [{Image src='img236741055943655201772.jpg' alt='' caption=''}]he following 3 rxns using the data provided. Calculate delta S and delta G for each of the three | Homework.Study.com](https://homework.study.com/cimages/multimages/16/img236741055943655201772.jpg)



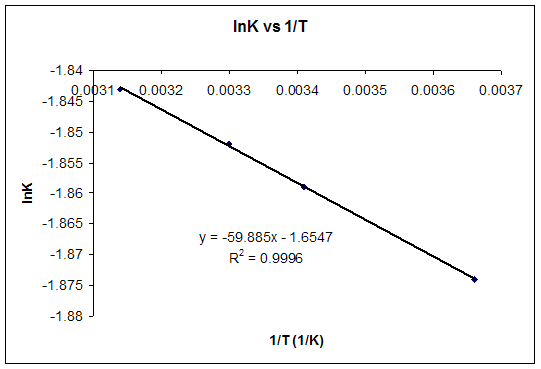



![15.2 Calculate the standard entropy change for a reaction [HL IB Chemistry] - YouTube 15.2 Calculate the standard entropy change for a reaction [HL IB Chemistry] - YouTube](https://i.ytimg.com/vi/IwRy4iYVQLI/maxresdefault.jpg)