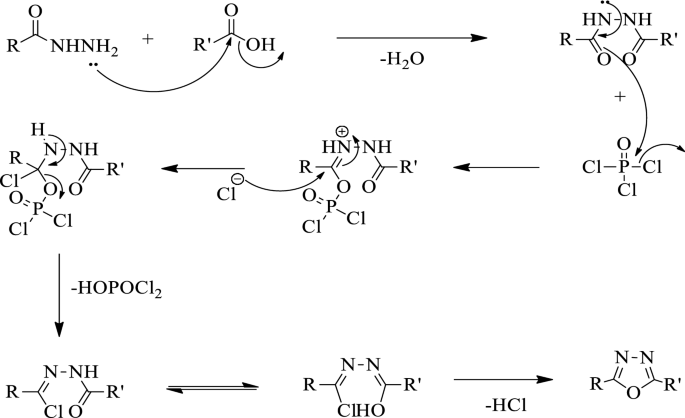
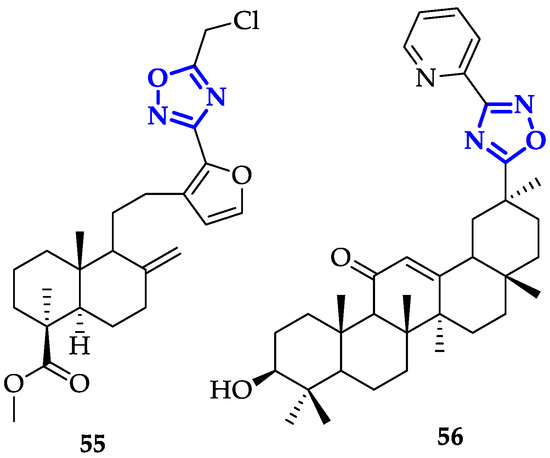
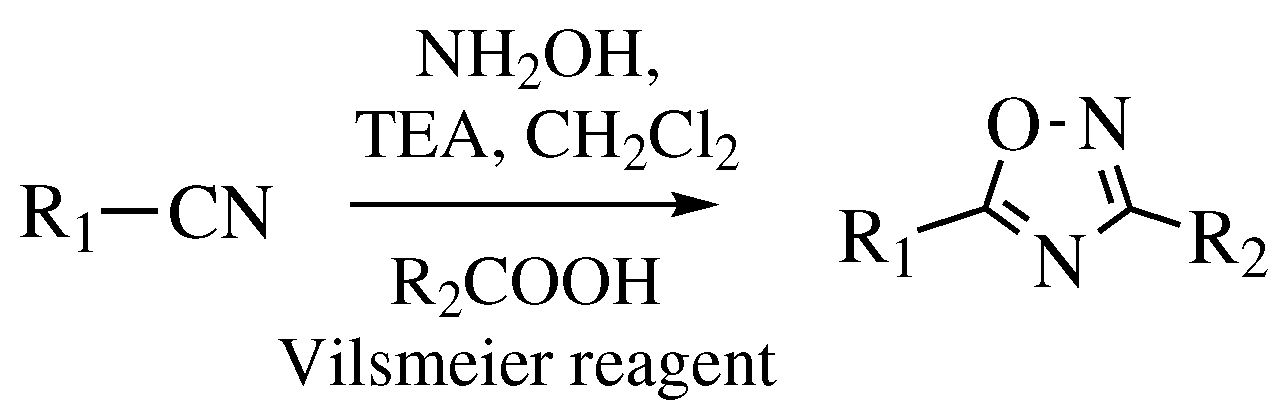Functionalization of 1,3,4-Oxadiazoles and 1,2,4-Triazoles via Selective Zincation or Magnesiation Using 2,2,6,6-Tetramethylpiperidyl Bases | Organic Letters

Cyclization and thiolation of chloroacetamide oxadiazole derivatives | Download High-Quality Scientific Diagram
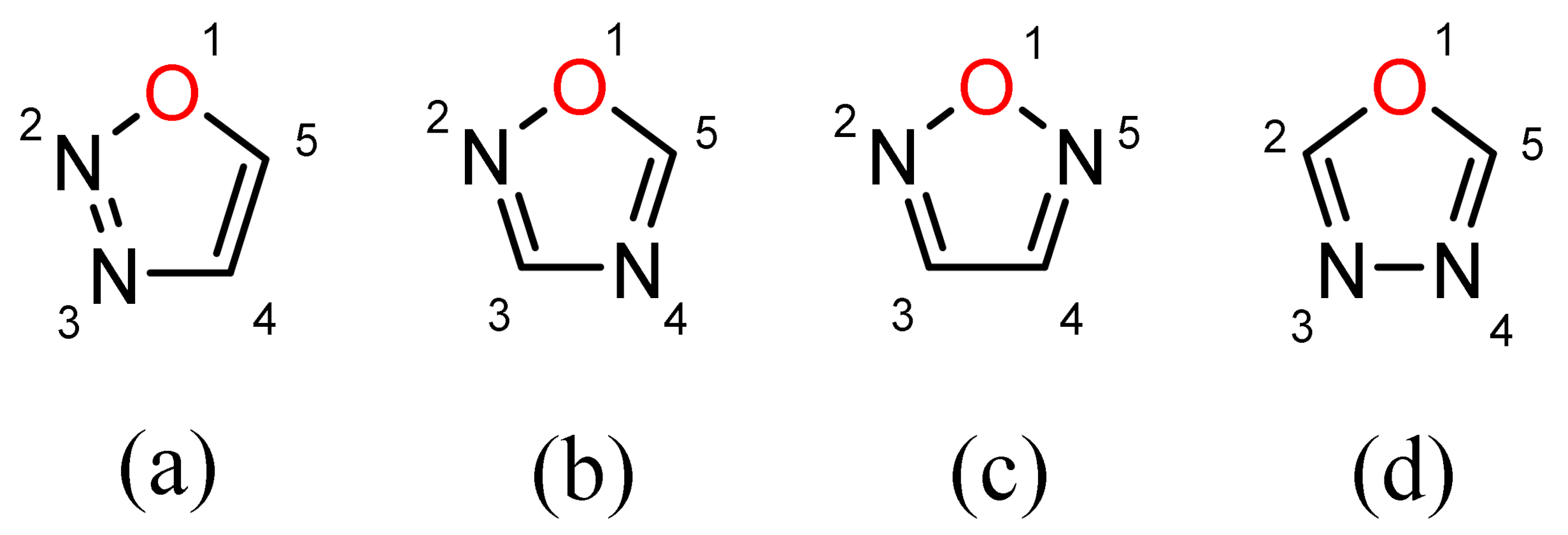
Applied Sciences | Free Full-Text | Arylation Reactions in the Synthesis of Biologically Important 2,5-Diaryl-1,3,4-Oxadiazoles | HTML

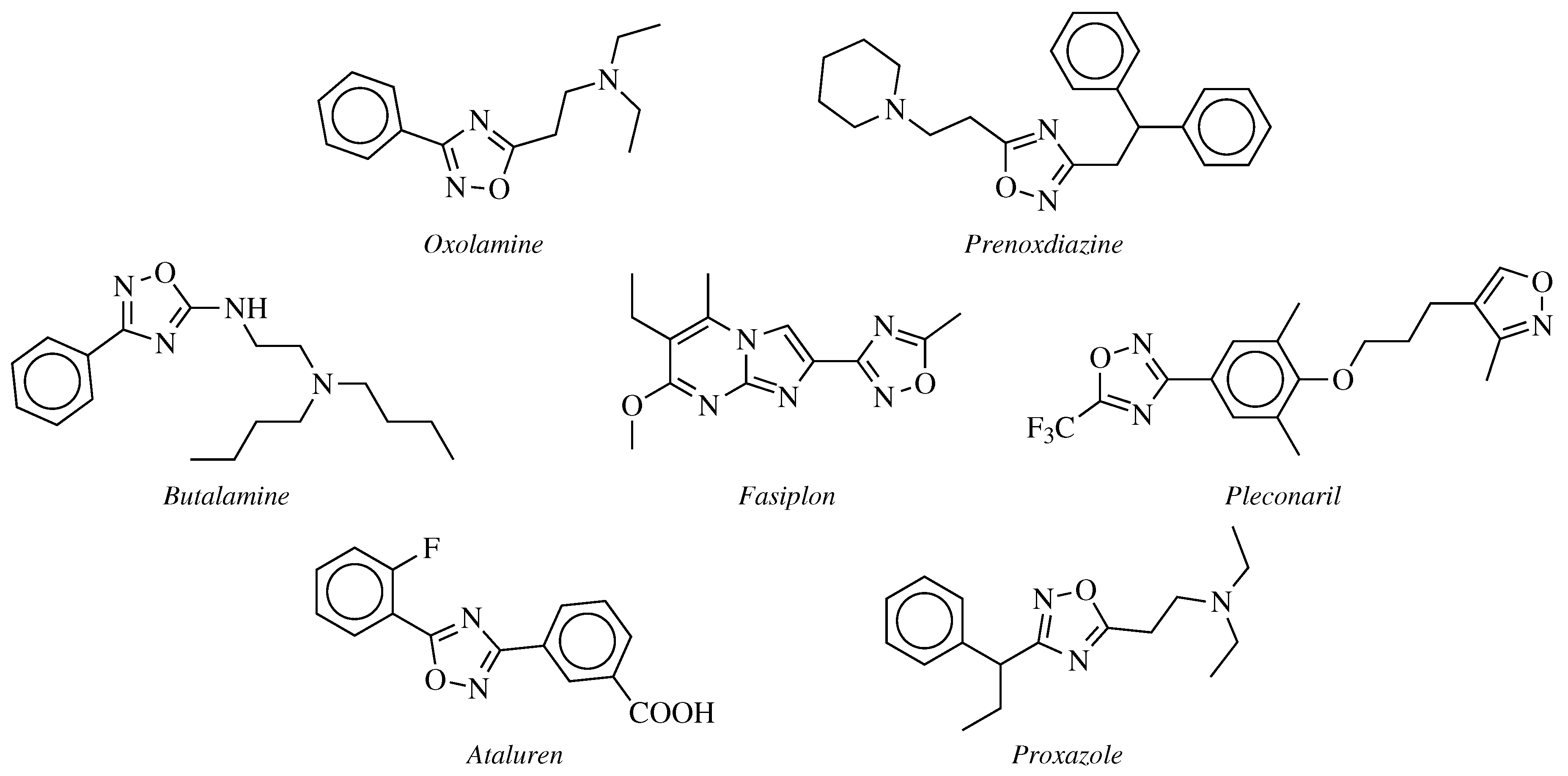
Development of 1,2,4-Oxadiazole Antimicrobial Agents to Treat Enteric Pathogens within the Gastrointestinal Tract | ACS Omega

Target Fishing Reveals a Novel Mechanism of 1,2,4-Oxadiazole Derivatives Targeting Rpn6, a Subunit of 26S Proteasome | Journal of Medicinal Chemistry

1,2,4‐ and 1,3,4‐Oxadiazole Synthesis by Palladium‐Catalyzed Carbonylative Assembly of Aryl Bromides with Amidoximes or Hydrazides - Andersen - 2014 - Advanced Synthesis & Catalysis - Wiley Online Library

Review on the Synthesis and Performance for 1,3,4‐Oxadiazole‐Based Energetic Materials - Du - 2021 - Propellants, Explosives, Pyrotechnics - Wiley Online Library

Development of 1,2,4-Oxadiazole Antimicrobial Agents to Treat Enteric Pathogens within the Gastrointestinal Tract | ACS Omega
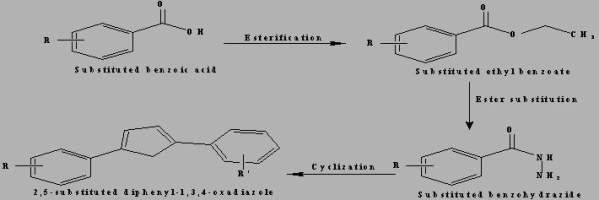
Synthesis and evaluation of substituted diphenyl-1,3,4-oxadiazole derivatives for central nervous system depressant activity | Organic and Medicinal Chemistry Letters | Full Text

Review on the Synthesis and Performance for 1,3,4‐Oxadiazole‐Based Energetic Materials - Du - 2021 - Propellants, Explosives, Pyrotechnics - Wiley Online Library

Synthesis and evaluation of 1,3,4-oxadiazole derivatives for development as broad-spectrum antibiotics - ScienceDirect

Novel Cytochrome P450-Mediated Ring Opening of the 1,3,4-Oxadiazole in Setileuton, a 5-Lipoxygenase Inhibitor | Drug Metabolism & Disposition

Dinitromethyl‐3(5)‐1,2,4‐oxadiazole Derivatives from Controllable Cyclization Strategies - Tang - 2017 - Chemistry – A European Journal - Wiley Online Library