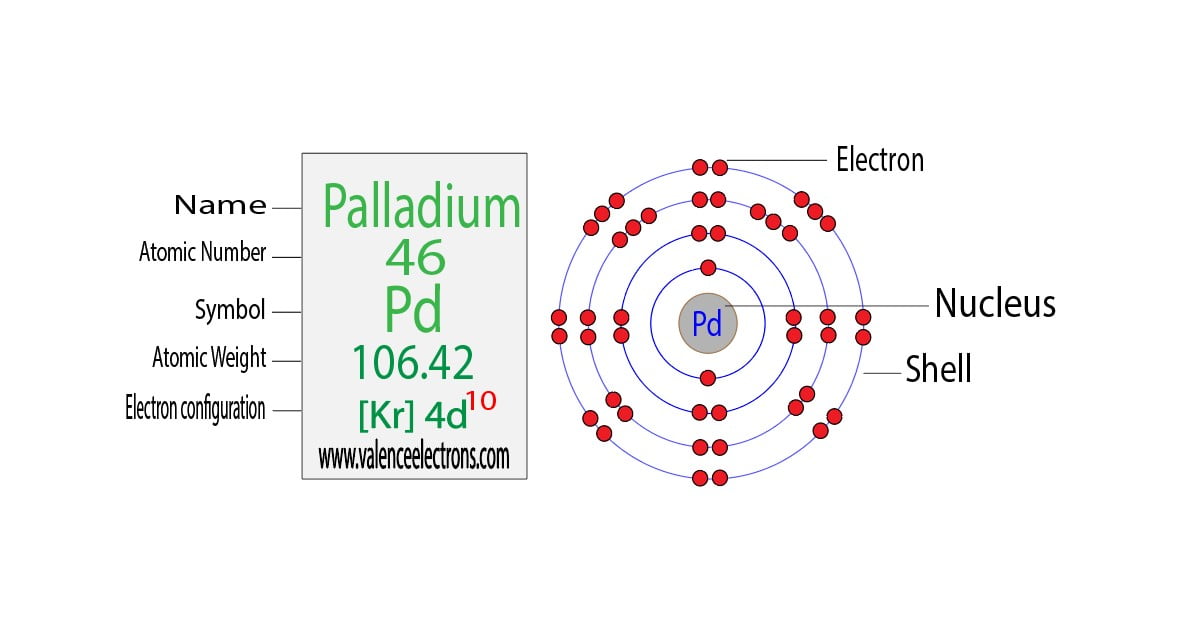
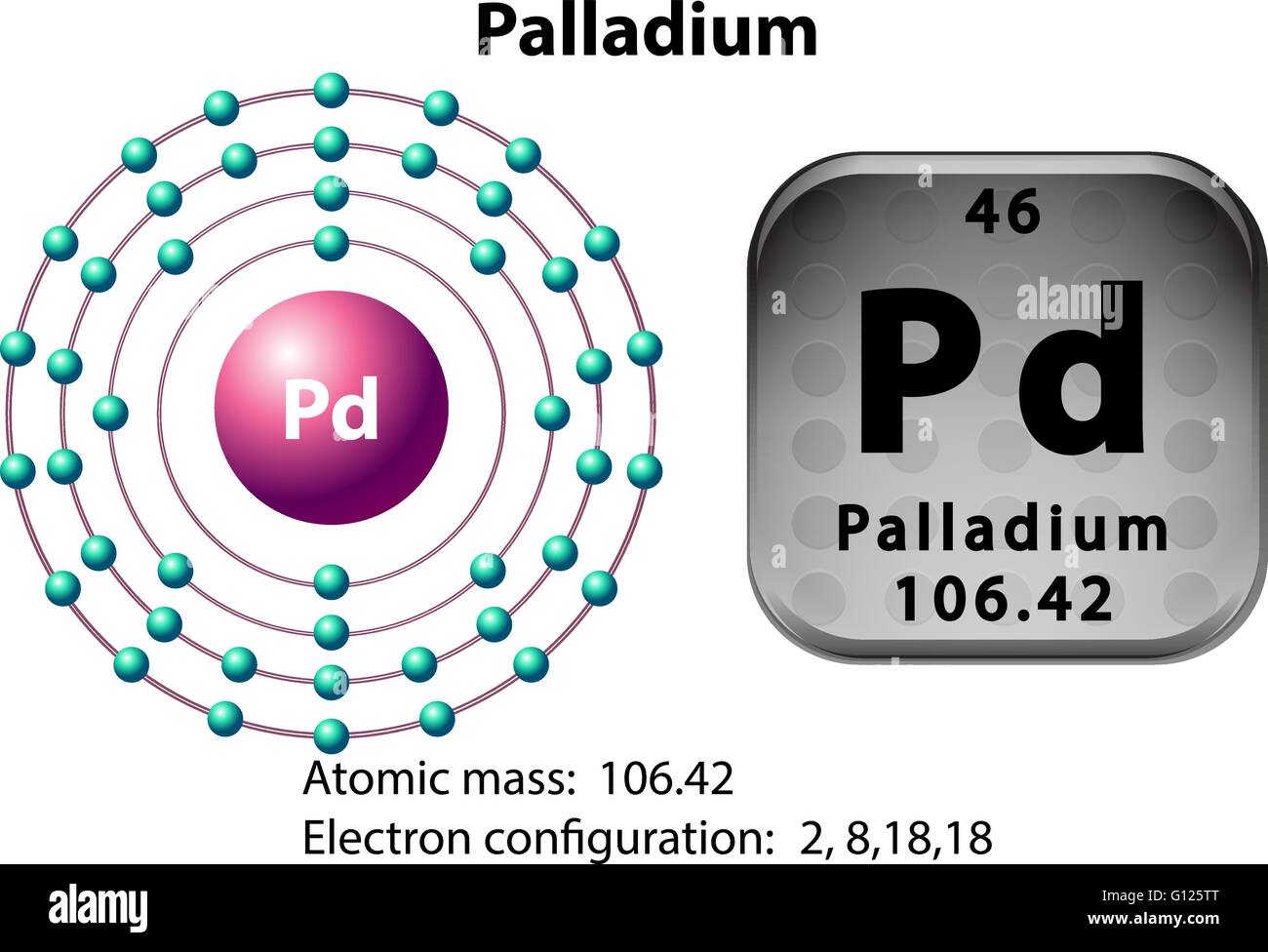
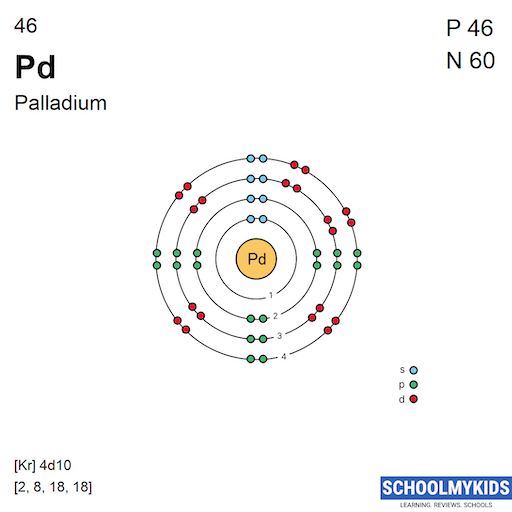
Why is palladium 5s0 4d10 more stable than 5s2 4d8, and why are the configurations of nickel and platinum different from palladium's? I had learned (and had always taught) that it is

Palladium (Pd; Z 46) is diamagnetic. Draw partial orbital diagrams to show which of the following electron - brainly.com



:max_bytes(150000):strip_icc()/Palladium-58b601c53df78cdcd83d0a00.jpg)







:max_bytes(150000):strip_icc()/aufbauexample-56a129555f9b58b7d0bc9f48.jpg)