
Pharmaceuticals | Free Full-Text | Synthesis, Antiproliferative Activity, and DNA Binding Studies of Nucleoamino Acid-Containing Pt(II) Complexes | HTML

Bis(imino)acenaphthene (BIAN)-Supported N-Heterocyclic Carbene Palladium Complexes with Ancillary Ligands: Readily Activated Precatalysts for Direct C–H Arylation of Thiophenes | Organometallics

Bis(imino)acenaphthene (BIAN)-Supported N-Heterocyclic Carbene Palladium Complexes with Ancillary Ligands: Readily Activated Precatalysts for Direct C–H Arylation of Thiophenes | Organometallics

PDF) Palladium hetero‐di(N‐heterocyclic carbene) complexes and their catalytic activities in direct C−H arylation of heteroarenes

Palladium(II) complexes of coumarin substituted 1,2,4–triazol–5–ylidenes for catalytic C–C cross–coupling and C–H activation reactions - ScienceDirect
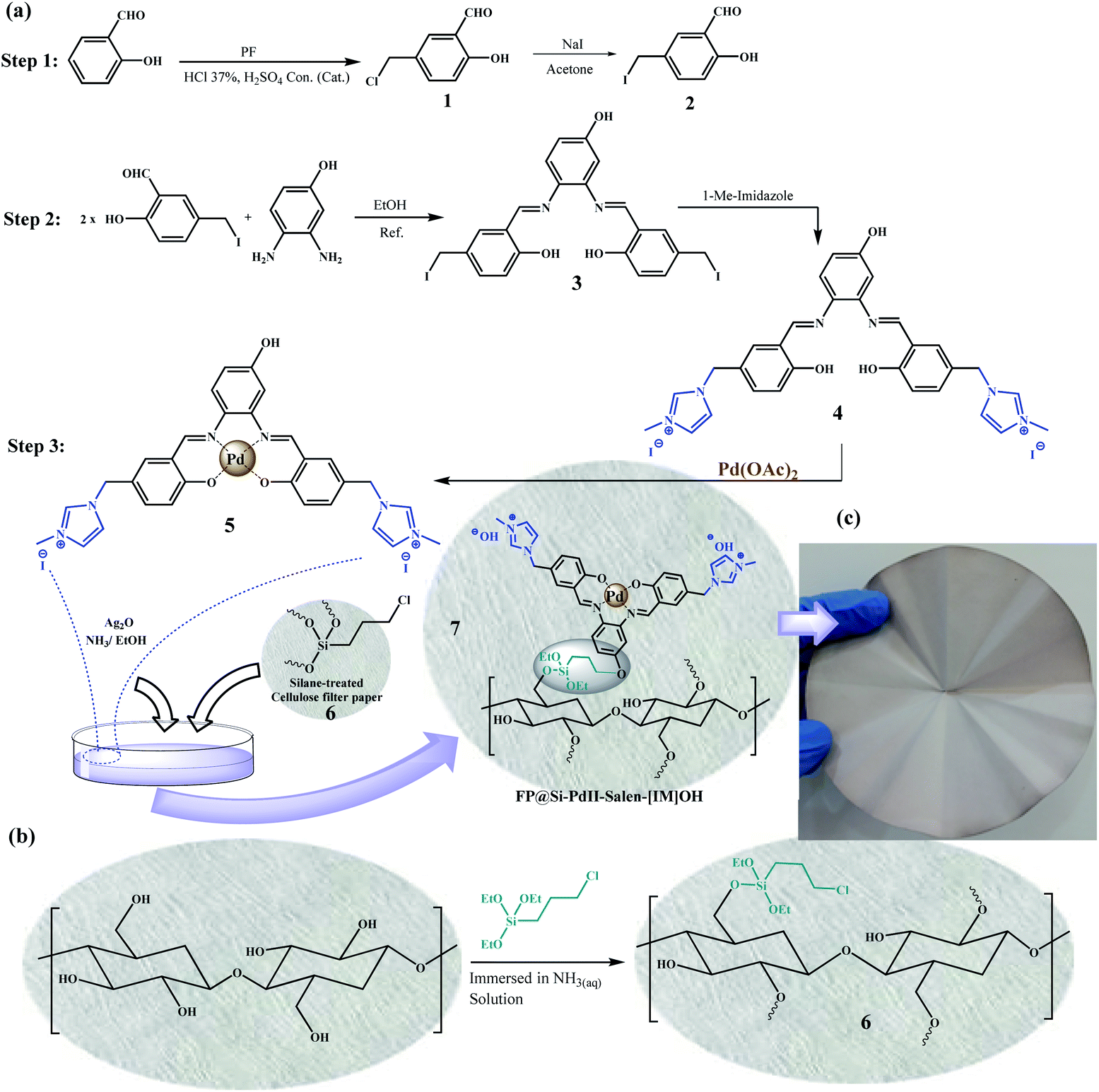
Catalytic filtration: efficient C-C cross-coupling using Pd (II) -salen complex-embedded cellulose filter paper as a portable catalyst - RSC Advances (RSC Publishing) DOI:10.1039/D2RA03440A

Palladium(II) complexes of coumarin substituted 1,2,4–triazol–5–ylidenes for catalytic C–C cross–coupling and C–H activation reactions - ScienceDirect

Palladium- and Platinum-Catalyzed Addition of Aldehydes and Imines with Allylstannanes. Chemoselective Allylation of Imines in the Presence of Aldehydes | Journal of the American Chemical Society

PDF) Palladium hetero‐di(N‐heterocyclic carbene) complexes and their catalytic activities in direct C−H arylation of heteroarenes
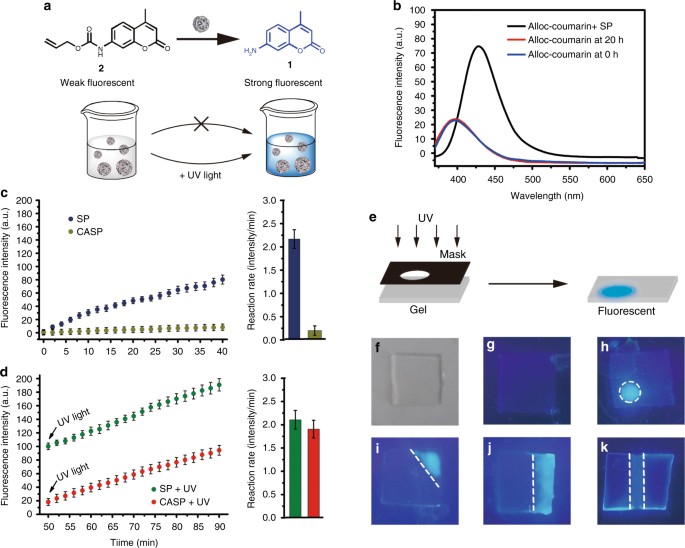
Designed heterogeneous palladium catalysts for reversible light-controlled bioorthogonal catalysis in living cells | Nature Communications
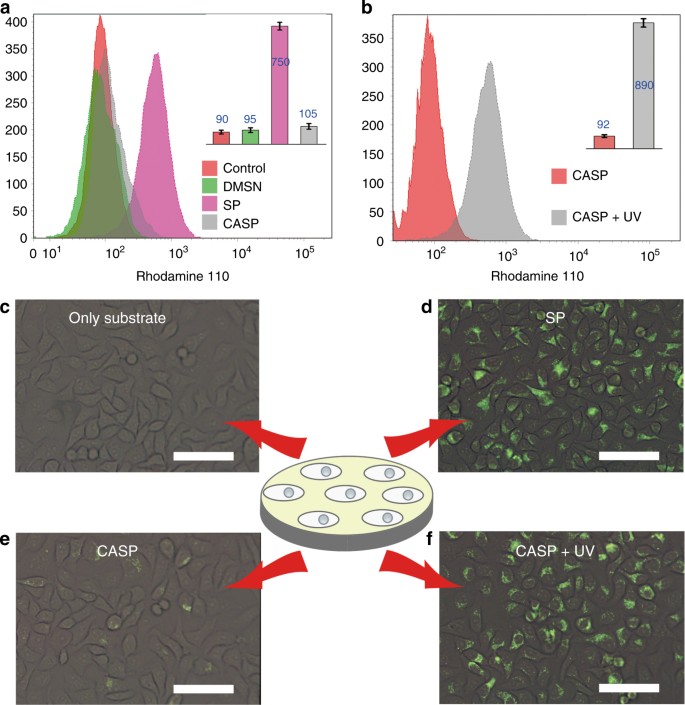
Designed heterogeneous palladium catalysts for reversible light-controlled bioorthogonal catalysis in living cells | Nature Communications

Palladium complexes of the N-fused heterocycle derived abnormal N-heterocyclic carbenes for the much-preferred Cu-free and the amine-free Sonogashira coupling in air - ScienceDirect

The kinetics and mechanism of polymer‐based NHC‐Pd‐pyridine catalyzed heterogeneous Suzuki reaction in aqueous media - Boztepe - 2019 - International Journal of Chemical Kinetics - Wiley Online Library

Revealing the Influence of Diverse Secondary Metal Cations on Redox‐Active Palladium Complexes - Golwankar - 2022 - Chemistry – A European Journal - Wiley Online Library

Di-Palladium Complexes are Active Catalysts for Mono-N-Protected Amino Acid-Accelerated Enantioselective C–H Functionalization | ACS Catalysis

Bis(imino)acenaphthene (BIAN)-Supported N-Heterocyclic Carbene Palladium Complexes with Ancillary Ligands: Readily Activated Precatalysts for Direct C–H Arylation of Thiophenes | Organometallics

Palladium complexes of the N-fused heterocycle derived abnormal N-heterocyclic carbenes for the much-preferred Cu-free and the amine-free Sonogashira coupling in air - ScienceDirect
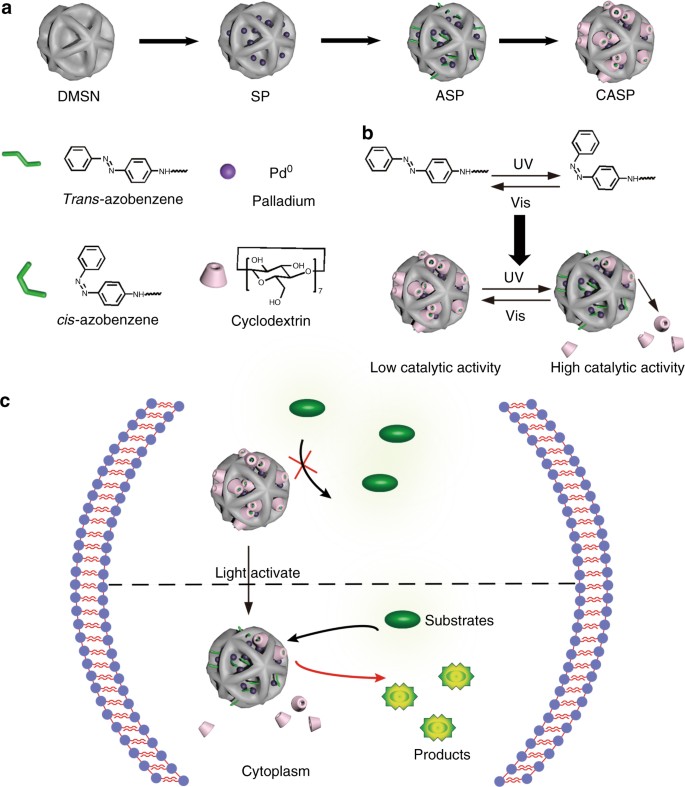
Designed heterogeneous palladium catalysts for reversible light-controlled bioorthogonal catalysis in living cells | Nature Communications








