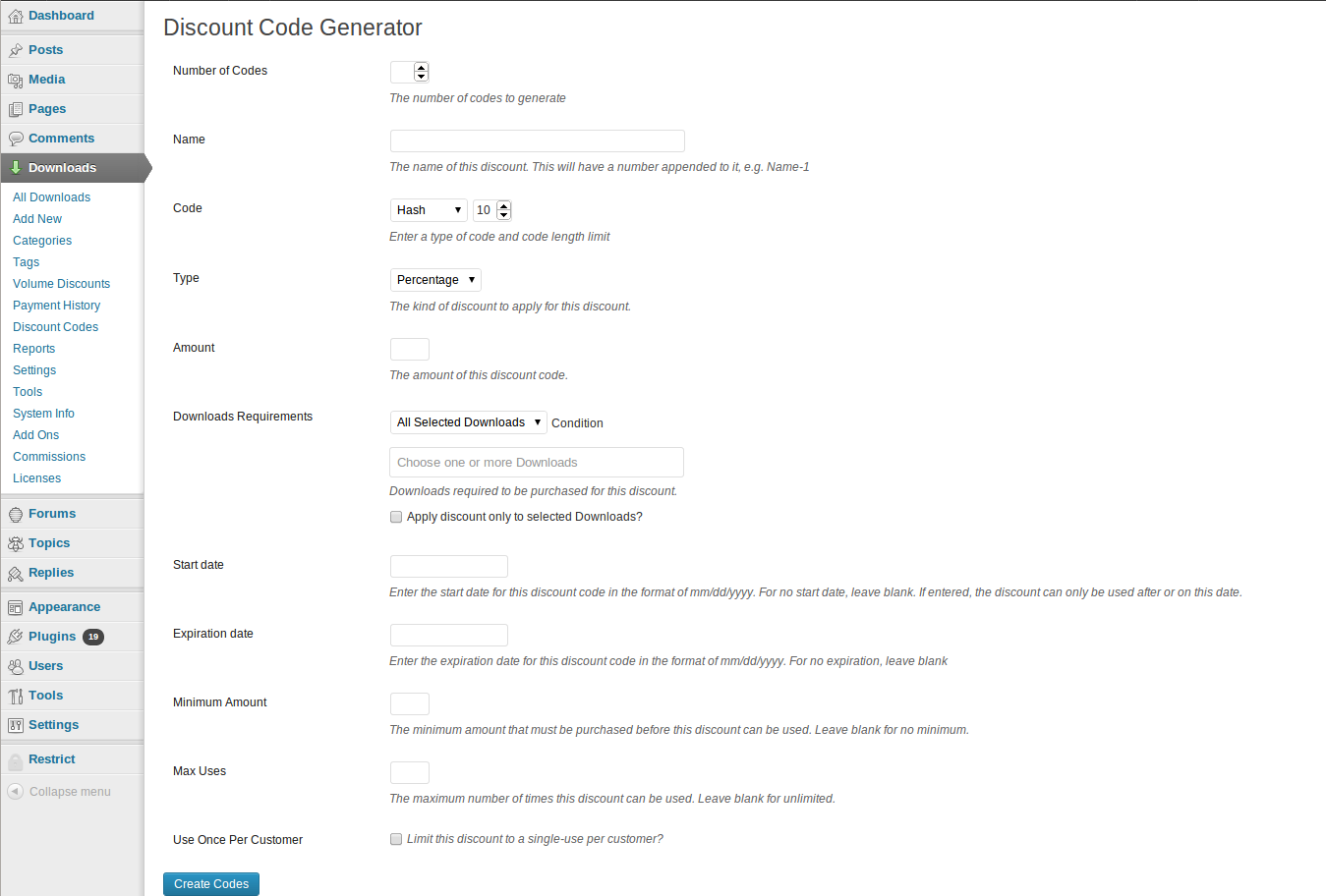
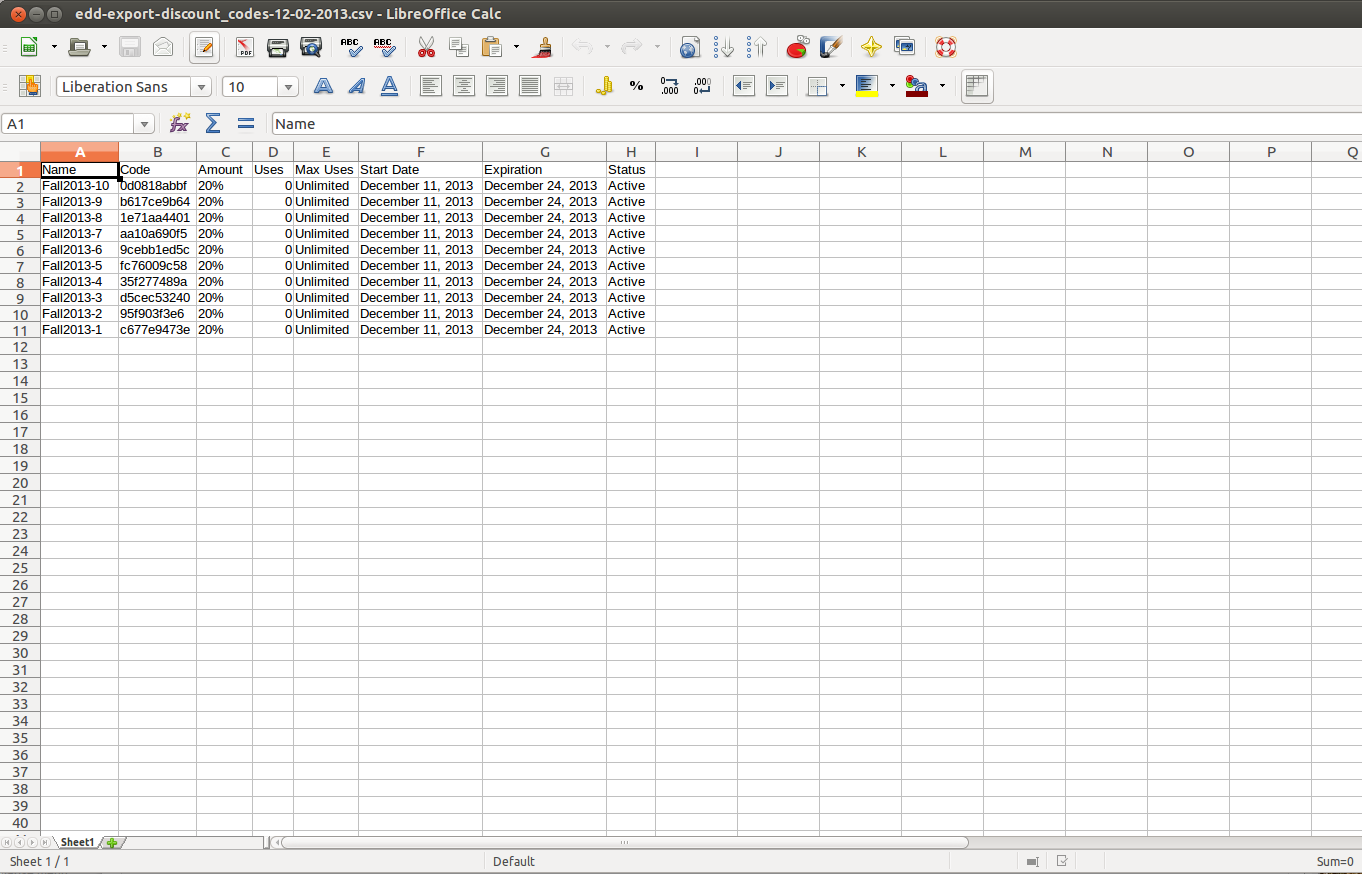
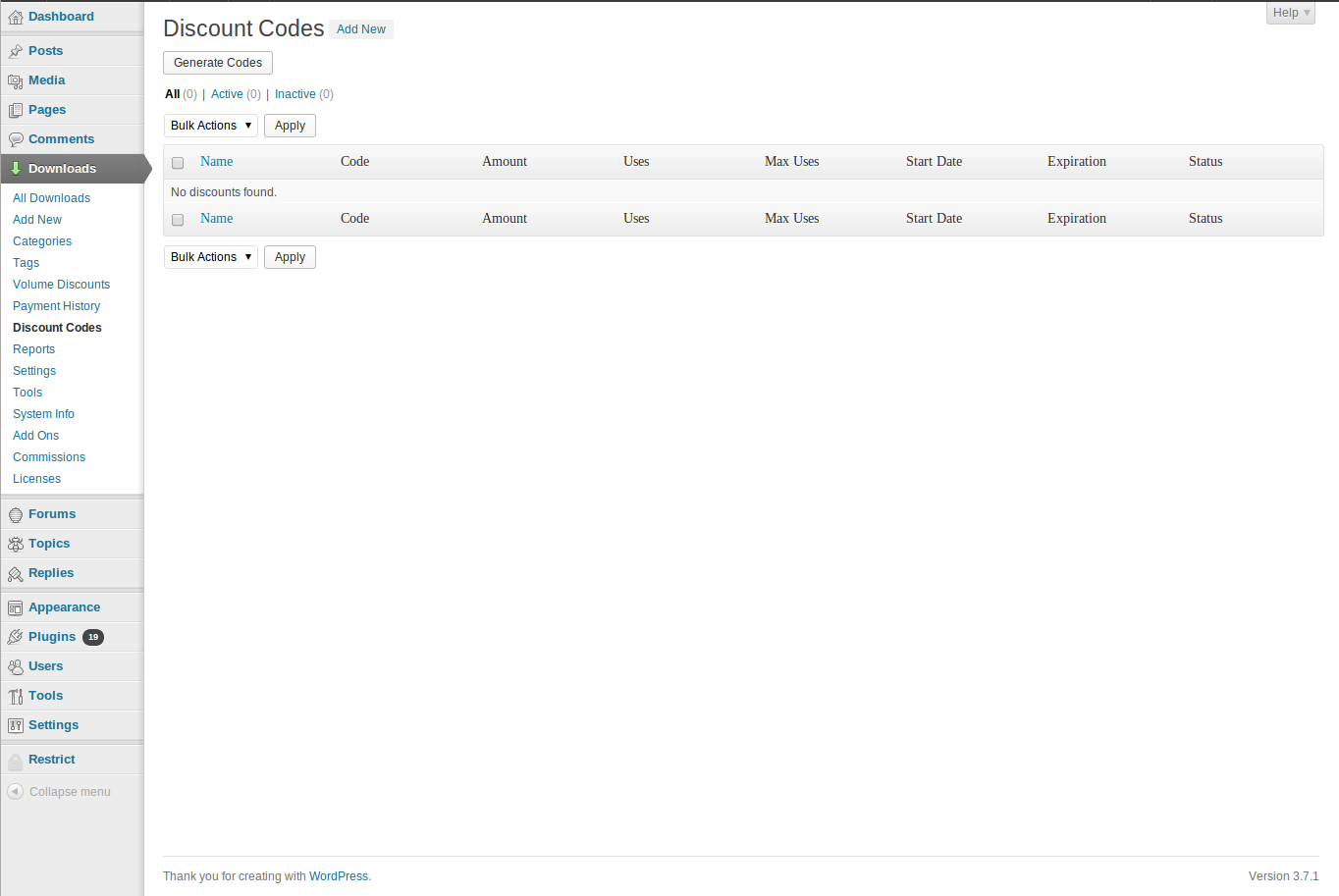
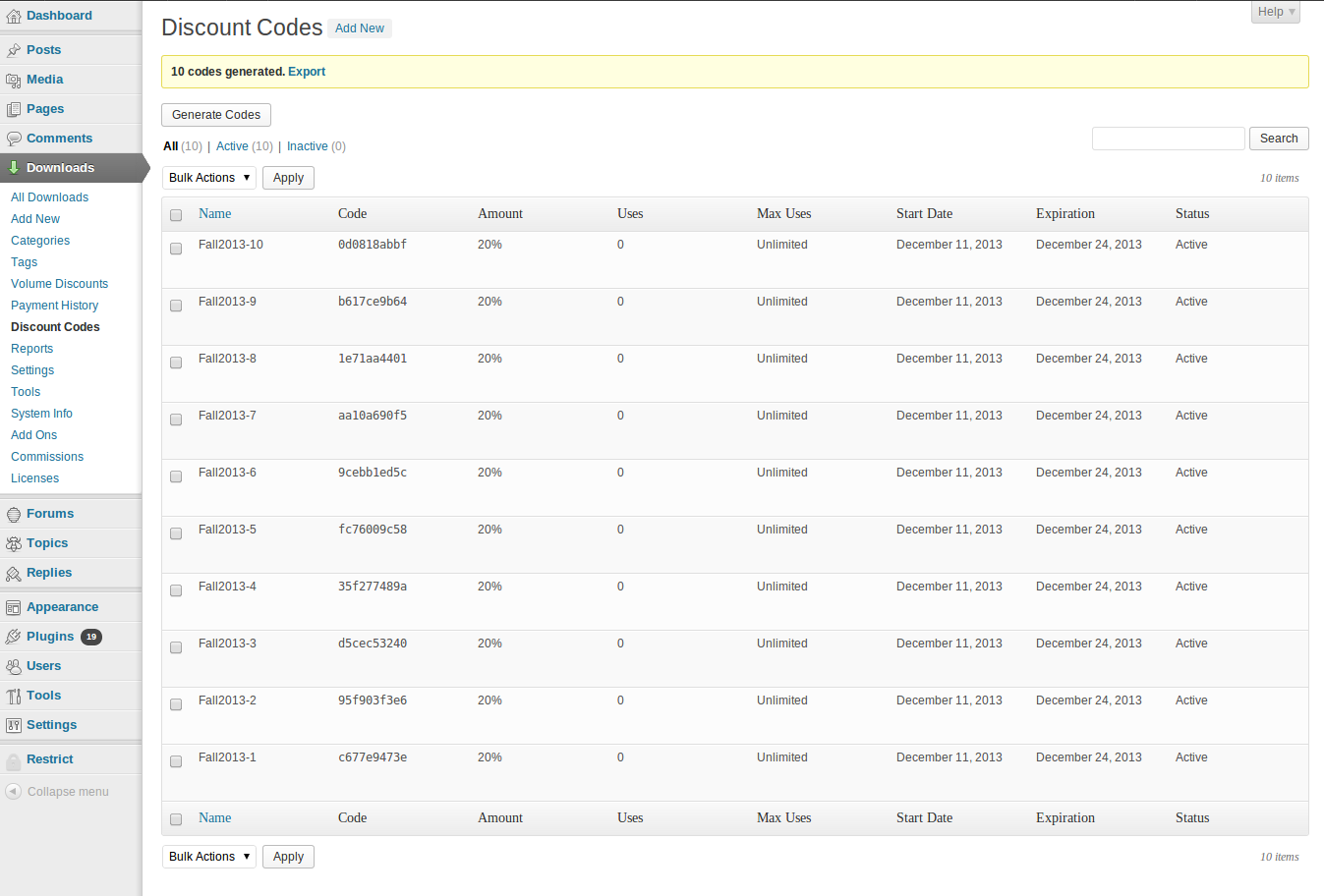
KNOWLEDGE TO LEAD | ISSN 2765-0626 | Volume 2 | Issue 1 | Oct. 2021 by Korean Educational Association for Empowerment of Softwar - Issuu

PDF) COVID-2019 Genome Sequence Analysis: Phylogenetic Molecular Evolution and Docking of Structural Modelling of Receptor Binding Domain of S Protein in Active Site of ACE2

Site-Specific Nonenzymatic Peptide S/O-Glutamylation Reveals the Extent of Substrate Promiscuity in Glutamate Elimination Domains | Journal of the American Chemical Society

DltC acts as an interaction hub for AcpS, DltA and DltB in the teichoic acid d-alanylation pathway of Lactiplantibacillus plantarum | Scientific Reports

Site-Specific Nonenzymatic Peptide S/O-Glutamylation Reveals the Extent of Substrate Promiscuity in Glutamate Elimination Domains | Journal of the American Chemical Society