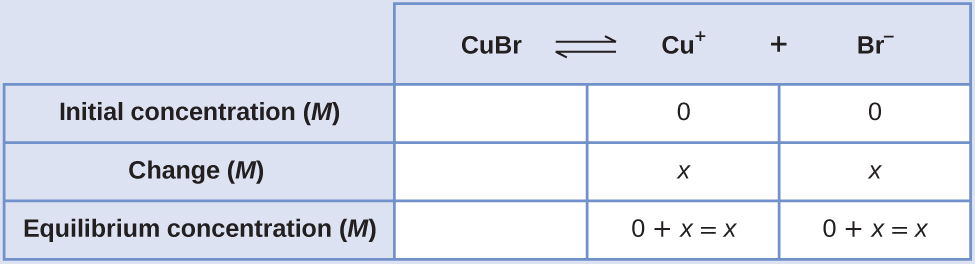
Table 1 from Solubility of amorphous Th(IV) hydroxide – application of LIBD to determine the solubility product and EXAFS for aqueous speciation | Semantic Scholar

Ksp SOLUBILITY EQUILIBRIUM. What is Ksp? Solubility product constant, last point of solubility Generally used for solutions which would be identified. - ppt download

Solubility Product Constant (Ksp) Overview & Formula | How to Calculate Ksp - Video & Lesson Transcript | Study.com

Table 2 from Temperature impact assessment on struvite solubility product: a thermodynamic modeling approach | Semantic Scholar

Table 2 from Temperature impact assessment on struvite solubility product: a thermodynamic modeling approach | Semantic Scholar
![SOLVED: Experiment 8 The Solubility Product Constant (Ksp) for a Sparingly Soluble Salt Table 2. Calibration Curve Data Standard Solution Volume (mL) 0.0024 M KzCrOa Total Volume (mL) [CrOz ]a (mol/L) Absorbance SOLVED: Experiment 8 The Solubility Product Constant (Ksp) for a Sparingly Soluble Salt Table 2. Calibration Curve Data Standard Solution Volume (mL) 0.0024 M KzCrOa Total Volume (mL) [CrOz ]a (mol/L) Absorbance](https://cdn.numerade.com/ask_images/c57c6ac42222486999c6aa956a0de286.jpg)
SOLVED: Experiment 8 The Solubility Product Constant (Ksp) for a Sparingly Soluble Salt Table 2. Calibration Curve Data Standard Solution Volume (mL) 0.0024 M KzCrOa Total Volume (mL) [CrOz ]a (mol/L) Absorbance
![SOLVED: (Concentration cell] concentration cell is set up determine the solubility product constant; Ksp"` of Pblz: In the anode of the concentration cell, solid Pblz added, which solublized sparingly. In the cathode. SOLVED: (Concentration cell] concentration cell is set up determine the solubility product constant; Ksp"` of Pblz: In the anode of the concentration cell, solid Pblz added, which solublized sparingly. In the cathode.](https://cdn.numerade.com/ask_images/12bbc11419d643199f5797066e273ce6.jpg)







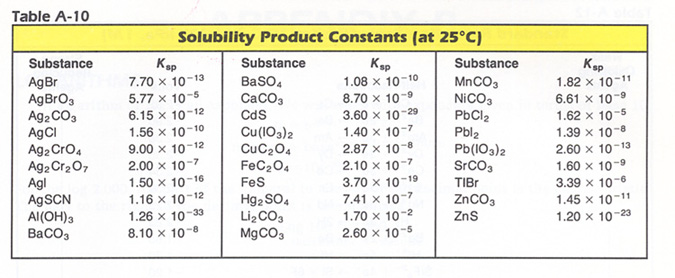
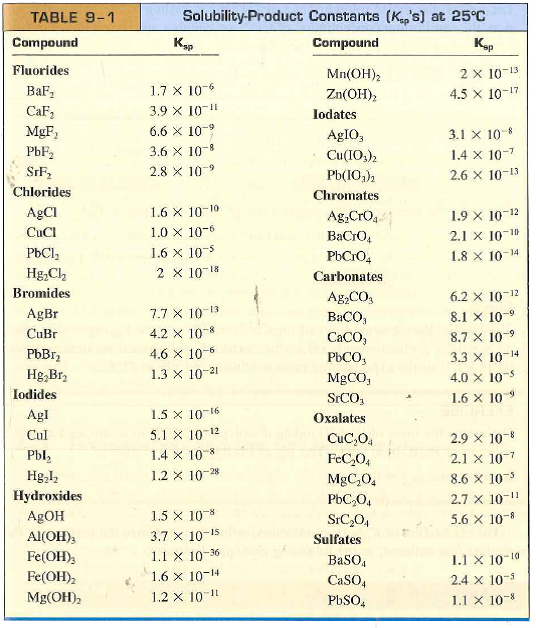

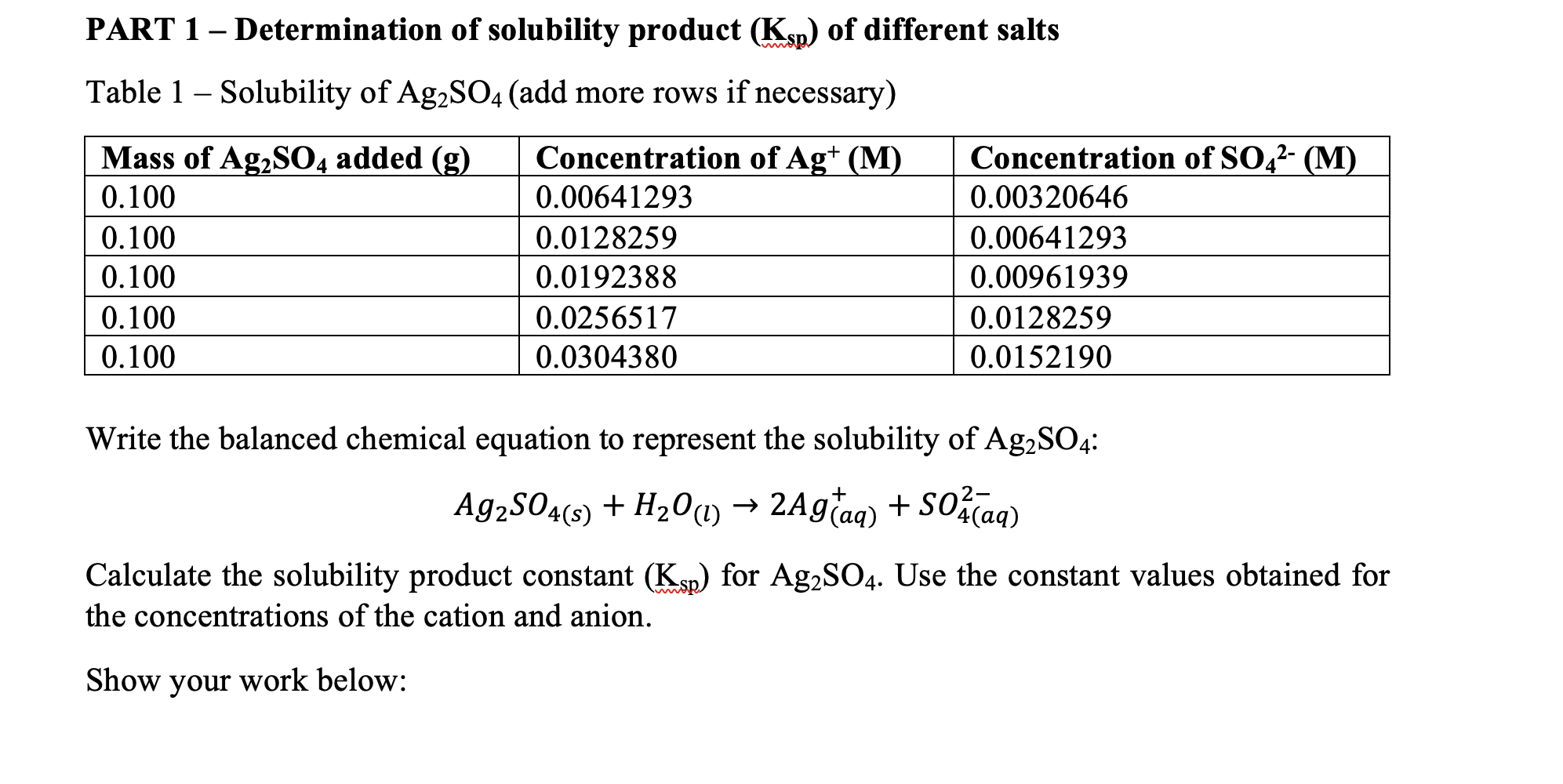

![PDF] THE SOLUBILITY-PRODUCT CONSTANT OF CALCIUM AND MAGNESIUM CARBONATES. | Semantic Scholar PDF] THE SOLUBILITY-PRODUCT CONSTANT OF CALCIUM AND MAGNESIUM CARBONATES. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3c09f4659498f1c291429e7e298bf43dcb7ebaaa/19-TableX-1.png)